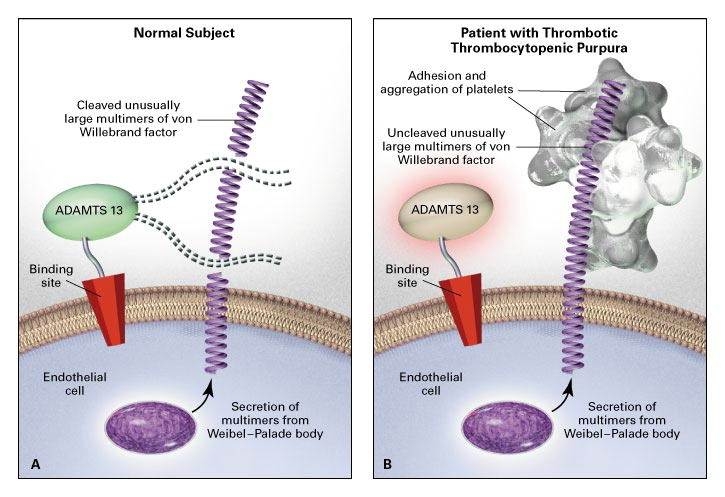
Figure 1.
Proposed Relation among the Absence of ADAMTS-13 Activity In Vivo, Excessive Adhesion and Aggregation of Platelets, and Thrombotic Thrombocytopenic Purpura.
In Panel A, in normal subjects, ADAMTS-13 (von Willebrand factor–cleaving metalloprotease) molecules attach to binding sites on endothelial cell surfaces and cleave unusually large multimers of von Willebrand factor as they are secreted by stimulated endothelial cells. The smaller von Willebrand factor forms that circulate after cleavage do not induce the adhesion and aggregation of platelets during normal blood flow. The ADAMTS-13 may use one of its thrombospondin-1–like domains or its arginine–glycine–aspartate (RGD) sequence to attach to the surface of endothelial cells.
In Panel B, absent or severely reduced activity of ADAMTS-13 in patients with thrombotic thrombocytopenic purpura prevents timely cleavage of unusually large multimers of von Willebrand factor as they are secreted by endothelial cells. The uncleaved multimers induce the adhesion and aggregation of platelets in flowing blood. A congenital deficiency of ADAMTS-13 activity or an acquired defect of ADAMTS-13 (such as that caused by autoantibodies or by a change in the production or survival of the protein) can lead to thrombotic thrombocytopenic purpura. Interference with the attachment of ADAMTS-13 to endothelial cells in vivo (for example, as a result of ADAMTS-13–receptor blockade by other types of autoantibodies) may also cause thrombotic thrombocytopenic purpura in patients with normal ADAMTS-13 activity in plasma.
From: Moake JL. Thrombotic microangiopathies. N Engl J Med 2002;347:587–600. Copyright © Massachusetts Medical Society. Reprinted with permission from Massachusetts Medical Society.