Chronic obstructive pulmonary disease, a disease that encompasses emphysema, chronic obstructive bronchitis, and small airway obstruction, is characterized by largely irreversible airflow obstruction.
10
It currently affects around 10% of the population over the age of 40 years and has recently become the third leading cause of death in the US.
11
,
12
The pharmacological treatment of COPD has generally consisted of bronchodilators. However, because of the presence of inflammation in COPD, inhaled corticosteroids, which had been shown to be highly effective for the treatment of asthma, were readily adopted in COPD in the 1980s despite the fact that no randomized controlled trials had yet evaluated their effectiveness in this indication.
The earliest randomized controlled trials to evaluate inhaled corticosteroids in the treatment of COPD were only published in the late 1990s. The first seven trials found no improvement in the decline of lung function over time and, except for the last two trials, found no reduction in exacerbation rates with various inhaled corticosteroids (ICS) compared with placebo, over periods ranging from 6 months to 3 years.
13
–
19
In the early 2000s, the next wave of randomized controlled trials all involved the evaluation of inhaled corticosteroids combined with a long-acting beta-agonist.
20
–
25
Most of these trials reported significant effects on lung function and reductions in exacerbation rates with the combination therapy, while the effects of inhaled corticosteroids alone were equivocal. Thus, the totality of these trials can be concluded to imply that any effectiveness of these medications is driven primarily by the long-acting beta-agonist component.
26
Despite this weak evidence or even evidence to the contrary, inhaled corticosteroids are prescribed to over 70% of COPD patients in the United States and Europe.
26
During this same period, several observational studies of large population-based cohorts, conducted using health care databases, were published. These studies, using a simplistic time-fixed definition of exposure, reported highly spectacular reductions in all-cause mortality of 30% to 40% with ICS use, alone or in combination with a long-acting beta(2)-agonist (LABA).
27
–
30
By using a time-fixed definition that does not allow drug exposure to vary over time, these studies introduced a bias known as “immortal time bias” that we describe in this observational study context.
31
–
35
Observational Study 1
To describe the role of immortal time bias in these studies, we use the first of these published studies.
27
This study used a cohort design to assess whether the use of inhaled corticosteroids after discharge from hospital for COPD was effective at reducing the risk of COPD readmission or all-cause death. All 22,620 patients over 65 years of age admitted to hospital for COPD in Ontario, Canada, between April 1992 and March 1997 were identified from this Province’s health insurance database. The patients were followed from the date of discharge for up to 1 year, or earlier if they were readmitted or died, in which case follow-up ceased at those points. The 11,481 patients who filled at least one prescription for an inhaled corticosteroid during the first 90 days after discharge were classified as users. The remaining 11,139 who did not were classified as non-users. An intent-to-treat analysis was performed on the basis of this classification using a proportional hazards regression model, accounting for several covariates. The resulting adjusted hazard ratio of all-cause death was found to be 0.71 (95% CI 0.65–0.78) for inhaled corticosteroid use relative to non-use, a 29% reduction.
Immortal time bias is introduced in this study by the definition of exposure in the cohort analysis. In this cohort study, a subject is considered exposed when an inhaled corticosteroid is dispensed at any time during the 90-day period after discharge. Hence, to be exposed, a patient must first survive the time until they receive that first prescription in that 90-day period. Thus, the time span between the date of discharge and the date of the first prescription of inhaled corticosteroids is called “immortal” because no deaths can occur during this period (Figure 1). More important, however, is the fact that subjects are classified as “users” of the drug during this immortal period even though the patient was not exposed until the first prescription was dispensed in that 90-day period. The misclassification of this time period as “exposed” when in fact it should have been classified as unexposed will engender immortal time bias. The solution is simply to use a time-dependent approach to data analysis that permits the patient to be classified as unexposed from cohort entry until the date of their first prescription, after which they can be classified as exposed. Methods based on person-time using Poisson models or more sophisticated techniques such as the Cox proportional hazards models with time-dependent exposure are available to account correctly for this problem.

|
Figure 1
Illustration of immortal time bias in the Sin and Tu observational cohort study of inhaled corticosteroids in patients discharged with COPD.
27
|
To illustrate the principle behind this bias, we used the simple person-time approach (estimating rate ratios with Poisson models to compute confidence intervals) on the data provided in the paper, after rounding the numbers for simplicity and making assumptions for unreported data. Thus, we considered that there were 12,000 patients per group, with a mean follow-up of 9 months, so that each group generated 9,000 person-years of follow-up, with 2,400 deaths occurring during follow-up, 1,000 in the ICS user group and 1,400 in the non-users. For the sake of illustration, we simply assumed that the mean delay between cohort entry (discharge) and the first ICS prescription among the ICS users was at 45 days, i.e. midway into the 90-day period used to define exposure. Table 1 shows that this would result in 1,500 immortal person-years of no ICS exposure misclassified as ICS exposed. The resulting rates of death for ICS users (1,000/9,000 = 11.1 per 100 person-years) and for non-users (1,400/9,000 = 15.6 per 100 person-years), based on these misclassified immortal person-years, produce a crude rate ratio of 0.71 (95% CI 0.66–0.77), which suggests a significant reduction in mortality. However, by properly reclassifying these 1,500 immortal person-years as unexposed, the rates would become 1,000/(9,000–1,500) = 13.3 per 100 person-years for ICS use and 1,400/(9,000+1,500) = 13.3 per 100 person-years for non-use, resulting in a corrected crude rate ratio of 1.0 (95% CI 0.92–1.08), suggesting no benefit whatsoever.

|
Table 1
Comparison between biased time-fixed data analysis and corrected time-dependent data analysis for the cohort study of inhaled corticosteroid (ICS) use and all-cause mortality in chronic obstructive pulmonary disease (COPD).
27
|
To illustrate further this bias with actual data from another cohort, we replicated the study using data from the computerized health care databases of Saskatchewan, Canada, to form the cohort of patients who were hospitalized for COPD between January 1, 1990 and December 31, 1997.
31
The cohort included 979 subjects, of whom 389 subjects either died or were re-hospitalized for COPD during the 1-year follow-up. During the first 90 days of follow-up, 39% were dispensed an inhaled corticosteroid. Using the same approach as Sin and Tu, namely the Cox proportional hazards models with time-fixed exposure, the hazard ratio was 0.69 (95% CI 0.55–0.86), suggesting a strong benefit with this drug. However, using the correct analysis with the Cox proportional hazards models with time-dependent exposure that properly classifies exposure as ICS non-use during the immortal time period and as ICS use only after the date of dispensing of the first ICS prescription, the hazard ratio becomes 1.00 (95% CI 0.79–1.26), suggesting no such benefit with ICS.
Observational Study 2
A variation of this bias was seen in another observational study of inhaled corticosteroids (ICS) in the treatment of chronic obstructive pulmonary disease (COPD), which claimed in its title to present “results from two observational designs free of immortal time bias.”
29
This claim turned out to be in fact erroneous and reflected a grave misunderstanding of immortal time bias. The authors identified, from the United Kingdom’s General Research Practice Database (GRPD), the cohort of all 4,398 patients aged 50 years and older hospitalized for COPD from 1990 to 1999. Cohort entry was taken as the date of discharge, with 1-year follow-up until readmission to hospital for COPD or death. Patients were considered exposed to ICS if they received a prescription of ICS on the same day of discharge. Using a propensity scores matched cohort analysis, the hazard ratio of COPD readmission or death associated with ICS use was 0.69 (95% CI 0.52–0.93), suggesting a significant 31% reduction in this outcome with ICS use.
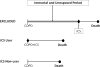
Immortal time bias is in fact introduced again with the definition of ICS exposure. It is stated that “treatment status was defined on the same day of discharge,” so that all 1,091 patients who were prescribed ICS on the day of discharge were correctly classified as ICS-exposed. However, of the remaining 3,307 patients, the non-users of ICS were incorrectly taken as merely the 538 patients “who were never exposed to ICS in their entire (one-year) follow-up period.” To comply with their stated Methods, they should have used all 3,307 patients from the cohort who were not prescribed ICS on the day of discharge. By excluding the 2,769 patients who were not prescribed ICS on the day of discharge but received an ICS later in the year of follow-up, the authors excluded a crucial component of follow-up time which is both unexposed and immortal, thus introducing a significant degree of immortal time bias in the results (Figure 2). Had the authors followed the correct method they described in the paper, namely to use “only patients whose treatment status was defined on the day of discharge,” they would have included all 3,307 such patients in the non-ICS group, and the rate ratio of COPD hospitalization or all-cause death with ICS would have been 1.48, not the reported 0.70.
33
|
Figure 2
Illustration of immortal time bias in the Kiri et al. observational cohort study of inhaled corticosteroids in patients discharged with COPD.
29
|
Observational Study 3
A further variation of this bias was seen in another observational study of ICS in COPD, also conducted using the GRPD. The cohort of 1,045 COPD patients treated with both an ICS (fluticasone) and a LABA (salmeterol) was compared with the cohort of 3,620 COPD patients who used other bronchodilators but not ICS or a LABA. The 3-year survival of the two cohorts was compared using survival analysis techniques. After adjusting for confounders, the combined users of ICS+LABA had a significant 52% lower mortality (hazard ratio 0.48; 95% CI 0.31–0.73), and the users of ICS only had a significant 38% lower mortality (hazard ratio 0.62; 95% CI 0.45–0.85) than the reference group of other bronchodilator users.
Immortal time bias is introduced in the hierarchical definition of exposure, where exposure is first assessed to identify the “exposed” cohort, namely those patients who received ICS+LABA. Only then was the “unexposed” reference group identified from the remaining patients as those who did not receive ICS or LABA, but only short-acting bronchodilators. However, many “exposed” subjects had used short-acting bronchodilators prior to their start of ICS+LABA, consistent with the stepped-care approach to COPD treatment. Thus, several subjects from the “exposed” group were in fact “unexposed” before switching to this exposure status. More importantly, however, this pre-exposure time during which subjects were “unexposed” is an immortal period since these subjects, in switching from the “unexposed” status to the “exposed” status, will necessarily do so alive. Had they died before switching, they would by definition have belonged to the unexposed group. Thus, the bias occurs because valid unexposed person-time of follow-up with no deaths is not accounted for in the reference rate of death. This results in an artificial increase in the rate of death of the reference group, leading to a spurious appearance of effectiveness. This bias was illustrated in another cohort of COPD patients, with the hazard ratio changing from a highly significant 0.66 (95% CI 0.57–0.76) to a non-significant 0.94 (95% CI 0.81–1.09) after properly accounting for this bias.
32
The TORCH Randomized Trial
In 2007, a large-scale randomized controlled trial was published, comparing an ICS+LABA (fluticasone+salmeterol) combination with placebo, LABA alone, or ICS alone, over a period of 3 years, on the primary outcome of death from any cause.
36
Of the 6,112 randomized patients, all-cause mortality was 12.6% in the ICS+LABA combination group, 15.2% in the placebo group, 13.5% in the LABA group, and 16.0% in the ICS group. The hazard ratio of death for the ICS+LABA combination compared with placebo was 0.82 (95% CI 0.68–1.00), while compared with ICS alone it was 0.77 (95% CI 0.64–0.93). Moreover, for ICS alone compared with placebo, the hazard ratio was 1.06 (95% CI 0.89–1.27). The authors concluded that the mortality reduction with combination therapy did not reach the predetermined level of statistical significance.
As these results were inconclusive, a further analysis of the data as a 2×2 factorial design of ICS (yes/no) and LABA (yes/no) was performed to improve the power and tease out the independent contribution of each component of the combination.
37
,
38
The interaction term to assess whether there is synergy between the two drugs was found to be non-significant (P = 0.32) suggesting that the combination of ICS and LABA is not particularly more effective than the two components added independently. Moreover, the factorial analysis showed that the LABA component is associated with a significant 17% reduction in mortality (RR 0.83; 95% CI 0.74–0.95; P = 0.0043), while the ICS component provides no reduction in mortality (RR 1.00; 95% CI 0.89–1.13; P = 0.99).
38
In essence, all observational studies suggesting a reduction in mortality with ICS use were shown to be flawed with immortal time bias, and proper re-analyses to avoid this bias eliminated any apparent protective effect of ICS.
31
,
32
,
34
,
35
In fact, Observational Study 2, described above, was specifically designed to emulate the TORCH randomized trial. It is now evident that the significant 38% and 52% potential reductions in mortality with ICS reported in this cohort study, in stark contrast with the absence of effects found in the TORCH randomized trial, were the result of immortal time bias.
