Vestibular schwannomas (VS)—also known as acoustic neuromas—are common benign cerebellopontine angle neoplasms, arising from the vestibular portion of the eighth cranial nerve (CN). Vestibular schwannomas occur with an incidence of 1:100,000 person-years.23,32,33 Vestibular schwannoma typically presents with ipsilateral hearing deterioration (affects 95% of patients), and tinnitus (affects 60% of patients). Additional presenting symptoms include dizziness, vertigo, trigeminal neuropathy (12%), facial neuropathy (5%), and rarely caudal cranial nerve involvement (IX–XII).34
Management of VS aims at eliminating brainstem compression and cranial nerve palsies secondary to tumor encroachment, with no functional worsening. Treatment options include observation (“wait and scan”), microsurgical resection through different avenues (discussed next), SRS, and fractionated conventional radiotherapy (FRT). Treatment is advocated in general, for those symptomatic or showing rapid tumor volume growths, regardless of age or comorbidities. A growth rate of more than 2.5 mm/year correlates to lower hearing preservation rates. In addition, conservative management is known to correlate to progressive hearing loss, averaging 2.77–5.39 dB/year.10
For nearly a century, surgical extirpation was the only option available for patients with VS. Technical and technique-related improvements over time resulted in dramatic improvements in surgical operative outcome measures, yet a GTR is still associated with a significant risk of related complications.35 The retrosigmoid surgical approach is associated with higher preservation rates of both hearing and facial nerve function for VS >1.5 cm in largest diameter. Still, this approach harbors higher rates of postoperative cerebrospinal fluid (CSF) leaks and headaches. The middle fossa surgical approach offers better outcomes for those with VS <1.5 cm in largest diameter. The translabyrinthine surgical approach in turn is reserved for patients with no functional hearing.10
Treatment of VS with SRS offers superior tumor control and facial nerve function preservation rates, with comparable hearing preservation rates. Current evidence supports the claim that SRS is associated with higher hearing and facial nerve function preservation rates and overall better functional outcomes. This is achieved with similar tumor control rates compared to microsurgical approaches. Stereotactic radiosurgery is currently considered the treatment of choice for VS <3 cm in largest diameter.10 For larger VS (>3 cm or 13 mL in volume), AHS should be considered as a safer option, with surgical debulking followed by SRS for the residual tumor as discussed next.
Tumor Volume Control
There is much controversy over complete versus incomplete removal of VS as the best management strategy. On the one hand, there is a clear proven correlation between the postoperative residual tumor volume and risk of progression or recurrence.
31 A recent report by Vakilian et al.
36 further supports this claim. A VS postsurgical volume >2.5 cm
3 showed recurrence in all cases in their report.
36 Reported rates of postsurgical facial nerve damage and hearing dysfunction remain significant.
37–40 On the other hand, tumor control rates of SRS (gamma-knife or LINAC-based) for small and medium-size VS are reported to reach as much as 97.5% of cases, combined with a 97% rate of facial nerve functional preservation and a median size decrease of 40% at 7 years post-SRS follow-up.
10,41 Thus, SRS allows for acceptable functional results in moderate-volume tumors and has emerged as the preferred upfront treatment alternative in such cases.
19,20,22
With the advances being made in all fields of medicine that improve quality of life, patients with large VS have higher expectations and lower tolerance and acceptance with regard to the functional outcome of VS; they expect and want similar functional outcomes as for those undergoing SRS for smaller-volume tumors in terms of neurological outcome and postoperative deficits. Tolerance for cosmetic and other complications such as facial nerve palsy has decreased dramatically. Such a complication is considered unacceptable nowadays and should be avoided at all costs.23 To date, reports on outcome for AHS (subtotal removal followed by adjuvant SRS) for larger-volume VS have been scarce and cohort sizes limited.42–48
In a recent meta-analysis by Rykaczewski et al.49 on SRS for VS (including AHS), which encompassed 28 studies (2007–2011) and 3,233 patients, mean tumor control of 92.7% was reported, at an average follow-up of 51.24 months.49 There is further support to the concept23,50,51 that AHS provides both excellent tumor control rates and desired preservation of facial nerve function.42–48,52,53
Facial Nerve Preservation
In microsurgery of large VS, the size of the tumor preoperatively serves as the key predictor for facial nerve preservation, both anatomically and functionally.
4,38 The incidence of surgically induced facial nerve palsy in those with VS >3 cm is 6-fold greater than in those with smaller lesions.
54
Reviewing available studies reporting a cranial nerve morbidity measure for those who received GTR versus STR for large-volume VS returns mixed results.29,55 Of note, most of the surgical series reporting postoperative facial nerve morbidity consider patients with House–Brackmann (HB) I–III as good results. However, from a functional and cosmetic point of view as well as quality of life, HB II–III is by no means normal function.31 Still, these results suggest that although surgeons prefer safety over GTR, STR in current practice (random, not as part of AHS) can still cause undue neurologic deficits without the added gain of GTR.31 However, STR of large VS has been reported to achieve excellent facial nerve function preservation rates in 80%–100% and serviceable (functional) hearing in up to 100% of patients.48 Pollock et al. performed a meta-analysis and compared postoperative facial nerve palsy after microsurgery and SRS, reporting it to be 19% and 1%, respectively.56
Although the influence of extent of resection on postoperative facial nerve preservation remains a matter of debate, the concept that keeping intra-operative mechanical stress during STR to a minimum might help reduce neural morbidity appears to be more widely accepted.23 Accordingly, even when facial nerve electrophysiological monitoring is used (as in most cases), some argue for termination of surgery before the facial nerve stimulation signal is lost.57 However, since the surgeon cannot accurately predict when this will happen,58,59 many argue for leaving tumor tissue abutting the nerve if the need arises. Better postprocedural facial nerve preservation rates have been achieved by AHS with STR, in the range of 82%–100% (HB I–II).42,46,60–62
Adaptive Hybrid Surgery for Large VS
One can approach STR for large VS using two different strategies. One consists of a planned STR in which the surgeon directs the surgical effort to preserve the cranial nerve and brainstem, resecting only the volume of tumor necessary for converting the residual volume to an ideal SRS target. This approach yields the best combined outcome, as recently reviewed by Iwai et al.
53 and Daniel et al.
31 A second approach consists of performing an STR (near-GTR), geared at leaving as little tumor as possible, which typically occurs at the level of the internal acoustic meatus. Since the facial nerve is particularly vulnerable at this location, this strategy has proven less favorable. The latter approach does not represent a real AHS approach.
Jeltema et al.63 reported a series of large-VS patients in which microsurgery was aimed at near-GTR with salvage SRS only when the residual volume showed growth. The authors reported normal facial nerve function (HB I) in 57.7%. In this study, 32.7% had mild (HB II and III) postoperative facial function dysfunction, and 9.6% had severe (HB IV and V) palsies. As discussed, this is not a true AHS approach, and most would argue for leaving a larger residual tumor in place to be followed by planned SRS. Concerning the timing of SRS after planned STR (as part of AHS), when larger VS residuals are left in surgery, many argue that complementary SRS should be performed in the months following surgery.31,53
Pan et al.62 compared two treatment approaches for large VS: AHS (group 1, n=18) versus GTR (group 2, n=17). Excellent facial nerve functional outcome (HB I and II) was noted in 89% and 35% in groups I and II, respectively. Hearing preservation was 100% and 0% in groups I and II respectively. Similar results were reported by Van de Langenberg et al.46 in a series of 50 patients. Brokinkel et al.50 published a recent systematic review analyzing six studies of AHS (gamma-knife radiosurgery following STR). A cohort of 159 patients with tumor diameters greater than 2 cm were reviewed. The average follow-up was 15 months, in which time excellent facial nerve function (HB I and II) was noted in 94%, and serviceable (functional) hearing preserved in 11.6%.
Daniel et al.31 reviewed 32 patients treated with AHS. Median follow-up was 24 months (range 4–78). Average presurgical tumor volume was 12.5 mL (range 1.47–34.9). Of note, in their series, Daniel et al.31 controlled the extent of surgical resection and capsular excision, progressing only until both superior and inferior borders of the facial nerve were identified surgically with electrophysiological neuro-monitoring. Thus, in a much more conservative surgical approach, a thin cuff of tumor capsule around the facial nerve course was intentionally left behind.31
Adaptive Hybrid Surgery Software
The adaptive hybrid surgery software (Brainlab AG, Munich, Germany(, approved by the US Food and Drug Administration in 2014,
64 simulates treatment plans (SRS, FRT) for postsurgical tumor volumes. The software assists in defining an SRS-ideal residual tumor volume (
Figure 2).
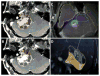 | Figure 2 Adaptive Hybrid Surgery Software (with Permission, from Brainlab Inc.) |
The AHS software allows the surgeon to define a realistic residual tumor target. Next, it formulates three separate radiation plans (possible approaches) for each tumor volume predefined by the surgeon for different complementary radiation tools (SRS, hypofractionated SRS, and FRT). The AHS software helps in balancing the risks of microsurgery (based on the surgeon’s initial delineation of a realistic residual tumor volume) against the risks of radiation-related toxicity to critical adjacent neurovascular structures from the SRS.65
Yang and his co-authors66 retrospectively compared target volume delineation defined manually by a surgeon to those formulated by the AHS software in seven patients with VS. The planned volumes were significantly smaller in the manual schemes as compared to those offered by the AHS software (1.6 mL versus 4.5 mL, P=0.004). The mean residual volumes were significantly smaller than the ideal volumes defined by AHS (2.2 mL versus 4.5 mL; P=0.02). As discussed, a smaller postsurgical residual volume translates to higher facial nerve damage rates and hearing loss.

 1,2* and Gil E Sviri, M.D., M.Sc.1
1,2* and Gil E Sviri, M.D., M.Sc.1