A 60-year-old man presented to the emergency room with jaundice and elevated hepatocellular and cholestatic liver enzymes in accordance with hypereosinophilia. The patient denied fever, abdominal pain, nausea, or vomiting. He described a pale stool and dark urine. He also denied consuming any drugs, herbal medicine products, or alcohol; he reported no personal or family history of liver or biliary disease, blood transfusion, or exposure to toxins. He denied a history of allergy, asthma, or recent travel.
On physical examination, the patient was alert, with prominent jaundice. He had signs of chronic liver disease: spider angiomas, palmar erythema, and clubbing. He had no ascites, gynecomastia, or testicular atrophy.
Laboratory findings are summarized in Table 1. Complete blood count showed hypereosinophilia, 3,636 eosinophils of 12,000 leukocytes; hemoglobin and platelets were within the normal range. Creatinine, urea, and electrolytes were normal. Total and direct bilirubin on admission were 13.5 mg/dL and 12.1 mg/dL, respectively. Liver enzymes were elevated in a combined pattern (hepatocellular and cholestatic) (Table 1). International normalized ratio (INR) was slightly elevated at 1.44, and partial thromboplastin time (PTT) was normal. Stool tests were negative for parasites.
 | Table 1 Blood Tests on Presentation. |
Serologic tests were negative for the following: hepatitis B surface antigen; anti-hepatitis B core immunoglobulin M (IgM); anti-hepatitis A virus IgM; anti-hepatitis C virus antibody; anti-Cytomegalovirus IgM; anti-Epstein–Barr virus IgM; anti-nuclear antibodies; anti-smooth-muscle antibodies; anti-mitochondrial antibodies; and anti-neutrophil cytoplasmic antibodies. Serum ferritin and ceruloplasmin levels were also normal.
Abdominal ultrasound performed on admission revealed hepatomegaly of 18 cm, lobulated edge, but no lesions were seen within the liver. Gallbladder and bile ducts were normal. A hyperechogenic non-homogenized lesion was seen at the head of the pancreas, 5.2 cm in diameter, which was suspected to be a space-occupying lesion.
Chest and abdominal CT demonstrated a suspected space-occupying lesion at the head of the pancreas, while liver, gallbladder, and spleen were normal. After further workup in the Department of Surgery, an endoscopic retrograde cholangiopancreatography (ERCP) was performed and did not show any abnormality. Both cytology and serologic tumor markers were normal. The patient developed post-ERCP pancreatitis which was managed conservatively with no complications. Following thorough investigation, the pancreatic lesion was concluded to be benign, and clinical follow-up was advised.
An incidental finding of a suspicious thyroid nodule was seen in the chest computed tomography (CT). Fine-needle aspiration for the nodule confirmed the diagnosis of papillary carcinoma. The patient had an elective total thyroidectomy and thyroxin replacement therapy was initiated.
At this point, a liver needle biopsy was performed (Figure 1, panels A–C), showing preserved lobular architecture (Figure 1A), mild portal inflammation with several eosinophils seen in the infiltrate (Figure 1B), focal bridging fibrosis (Figure 1C), and revealing features of chronic active hepatitis and marked eosinophilic infiltration with bridging fibrosis.
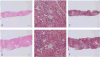 | Figure 1 Liver Biopsy at First Evaluation and Repeated Biopsy Four Years Later. |
Bone marrow biopsy excluded lymphoproliferative disease, and a diagnosis of idiopathic hypereosinophilic syndrome with eosinophilic hepatitis was established. Following discharge, the patient attended our Liver Unit for follow-up.
At this stage, he remained asymptomatic with marked eosinophilia and abnormal liver function test. Despite being asymptomatic, and due to the risk of progressive liver function, treatment with systemic corticosteroids (prednisone 40 mg/d) was started. Three weeks later, a marked drop of eosinophilic count to a normal level was seen, in accordance with normal levels of bilirubin and normal hepatocellular enzymes, while the cholestatic liver enzymes remained elevated.
A slow tapering down of prednisone over 2 months was initiated; however, a rapid rise of peripheral eosinophilia (24%) was noticed when reaching the lowest dose of prednisone 10 mg/d, along with elevated liver enzymes and bilirubin. Following this, prednisone 40 mg/d was restarted, with improvement of eosinophilic counts in two weeks. However, minimal improvement of liver enzymes was seen during this time.
Maintenance therapy on corticosteroids was continued for several months with clinical and laboratory response. Due to the corticosteroids therapy, he developed diabetes mellitus, which necessitated insulin therapy.
Several attempts at systemic steroid tapering down resulted in recurrent liver enzymes elevations with the lowest effective dose of 10 mg. Thus, resuming prednisone 40 mg/d along with azathioprine 75 mg/d was started, with later slow tapering down of corticosteroids.
Normal liver function study and eosinophilic count were achieved on prednisone 15 mg/d and azathioprine 75 mg/d (Figure 2). Further prednisone tapering down to the lowest effective dose of 5 mg/d and raising azathioprine to 100 mg/d was done.
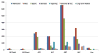 | Figure 2 Eosinophilic Count, Liver Function Tests, and Enzymes from Presentation to Date. |
Bilirubin and liver enzymes were normalized, as were albumin and INR. For histopathological assessment, a repeat liver biopsy (Figure 1, panels D–F) was done and showed portal inflammation (Figure 1D) with fewer eosinophils in the infiltrates (Figure 1E) and no change in fibrosis (Figure 1F, trichrome).
After 16 years of follow-up, the patient reported regular daily activity on chronic medications which included azathioprine 100 mg/d, prednisone 5 mg/d, thyroxin 150 μg/d, atenolol 25 mg/d, hydrochlorothiazide 12.5 mg/d, and ranitidine 40 mg/d.

 1* Johad Khoury, M.D.,2* Yaniv Zohar, M.D.,3 Afif Yaccob, M.D.,1 Ella Veitsman, M.D.,1,4 and Tarek Saadi, M.D.
1* Johad Khoury, M.D.,2* Yaniv Zohar, M.D.,3 Afif Yaccob, M.D.,1 Ella Veitsman, M.D.,1,4 and Tarek Saadi, M.D.