Breast cancer is the most commonly diagnosed malignancy and the second most common cause of cancer-related mortality among women in the United States.1 Current guidelines recommend screening for breast cancer with early mammography. Additional breast ultrasound and/or magnetic resonance imaging (MRI) are recommended depending on the patient’s age and existing risk factors.2,3 Screening programs allow early diagnosis and, therefore, have been shown to improve patient outcomes and reduce mortality.4
Treatment of early breast cancer is complex and involves a combination of surgery, radiotherapy, and, in selected cases, systemic treatments (chemotherapy, biological and hormonal therapy). The choice of treatment strategy should be based on the tumor burden/location and biology, as well as the age, menopausal status, general health status, and preferences of the patient.5
Breast cancer preoperative workup includes basic blood tests, breast and axillary ultrasonography, bilateral mammogram, and biopsy. Consideration of additional imaging for systemic staging including chest and abdominal computed tomography (CT), bone scan, and PET-CT is recommended only in the presence of signs and/or clinical symptoms of metastatic disease.5 The current guidelines recommend using PET-CT only in stage IV or recurrent disease. In addition, the use of PET-CT is considered optional in current guidelines in patients with lymph node involvement or HER2-positive disease.5 Therefore, no clear recommendation for PET-CT in patients with early breast cancer exists. Moreover, the current guidelines do not recommend considering the use of PET-CT in search of second primary malignancies in breast cancer patients.
The rapidly growing availability of fluorodeoxyglucose (FDG) positron emission tomography (PET)-CT scan presents a relatively new diagnostic possibility and has thus led to changes and modifications of indications for its use over the years. The PET-CT scan is a useful test in different oncology indications, including detection of occult primary malignancy in patients with metastatic disease, cancer staging, assessment of treatment response, and detection of disease recurrence and/or progression.6 However, the role of PET-CT in early breast cancer remains unclear, and evidence regarding the advantages of its routine use for initial staging of breast cancer is limited.7
Several studies have reported PET-CT to be of no practical value in patients with early breast cancer, (e.g. tumors smaller than 2–3 cm in patients with no palpable nodes).8–11 Therefore, currently, routine use of PET-CT is not supported by accepted guidelines.5
Nevertheless, PET-CT has been suggested to be of value in the detection of second primary malignancies in patients with breast cancer.12 Second primary malignancies are classified as either synchronous or metachronous. Synchronous second primary malignancies are tumors that occur within 6 months of the diagnosis of the first malignancy, whereas metachronous malignancies are those which develop 6 months or more after the diagnosis of the primary malignancy.12,13
Several studies have shown that women with breast cancer have a higher risk of developing a second primary malignancy as compared to the general population.14,15 Different factors may contribute to the development of secondary primary malignancies, such as previous chemotherapy or radiotherapy, young patient’s age, and hormonal manipulations during adjuvant treatment.16,17 Most studies have examined the association between treatment methods such as radiotherapy and chemotherapy, and development of second primary malignancy.16,17 The survival for breast cancer patients with second primary malignancy is significantly poorer.16
In this report, we present a case series of patients with non-metastatic breast cancer who underwent a PET-CT scan which revealed a synchronous, aggressive, second primary malignancy.
Case 1
A previously healthy, asymptomatic 58-year-old woman with no family history of malignancy was evaluated with routine screening mammography. The mammography revealed a 3 cm field of micro-calcifications in her right breast. On physical exam, no palpable mass or enlarged axillary lymph nodes were noted. Breast ultrasonography also revealed no findings. The patient underwent a core biopsy, which was consistent with ductal carcinoma
in situ (DCIS). The patient completed the accepted preoperative assessment, and, upon her request, PET-CT was performed. The PET-CT scan revealed a 5 cm mid-rectal mass, which was later biopsied (
Figure 1). A diagnosis of rectal adenocarcinoma was made, and a full workup was performed, after which the rectal tumor was defined as locally advanced (T4N0M0). The patient received a course of preoperative chemo-radiotherapy and anterior resection, followed by a right lumpectomy.
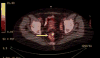 | Figure 1 A PET-CT Scan Showing a Rectal Mass. |
Case 2
A 67-year-old woman with a medical history of impaired fasting glucose and dyslipidemia was under follow-up due to a history of breast cancer. The patient had a history of breast cancer in her left breast which was treated with a left lumpectomy followed by chemotherapy and radiotherapy. Eleven years after the initial diagnosis, the patient had a recurrence and was treated with mastectomy and adjuvant chemotherapy. Genetic screening for BRCA was negative. During follow-up, 10 years after the mastectomy, routine mammography demonstrated a 1.4 cm mass in her right breast. Ultrasonography revealed a 1.3 cm mass on the external upper quadrant of the right breast, with no pathologic axillary lymph nodes. A biopsy confirmed the diagnosis of infiltrating ductal carcinoma, ER +3, PR +3, HER2 +2 (FISH negative), Ki-67 40%. The patient had no clinical signs of metastatic disease. The patient completed a PET-CT scan which revealed a heterogeneous large pelvic mass, 9×12 cm. The mass was biopsied, and the results were consistent with a gastrointestinal stromal tumor (GIST). Following a multidisciplinary team discussion, the patient underwent a simultaneous operation on both breast and GIST, which required a segmental small bowel resection. After completion of the abdominal procedure, a right breast lumpectomy with sentinel lymph node biopsy was performed.
Case 3
A 59-year-old woman with a history of Meniere’s disease and no family history of malignancy was evaluated with routine screening mammography that revealed a suspicious 12 mm mass on her left breast. On breast sonography, a 12 mm irregular mass was identified with no pathologic axillary lymph nodes. On biopsy, the mass was found to be invasive ductal carcinoma grade 2–3, ER +3, PR +3, HER2 negative, Ki-67 index 10%. The patient was further evaluated with a breast MRI and a PET-CT scan. The MRI demonstrated a 19 mm mass on the left breast without axillary lymphadenopathy, and PET-CT revealed a sub-pleural 12 mm mass in the right upper lobe of the right lung. Biopsy from the pulmonary mass was consistent with squamous cell carcinoma of the lung with clinical staging of T1N0M0. After a multidisciplinary discussion, it was decided to operate on the breast first. The patient underwent a lumpectomy and sentinel lymph node biopsy. Following the breast procedure, the patient underwent a thoracoscopic lobectomy of the right lung.
