|
|
|
|
RMMJ
Rambam Maimonides Medical Journal
Rambam Health Care Campus
2012 July; 3(3): e0015.
ISSN: 2076-9172
Published online 2012 July 31.
doi: 10.5041/RMMJ.10086
Copyright: © 2012 Novak A, et al.
Modeling Catecholaminergic Polymorphic Ventricular Tachycardia using Induced Pluripotent Stem Cell-derived Cardiomyocytes
Atara Novak, M.Sc.,1,
2,
3 Avraham Lorber, M.D.,3,
4 Joseph Itskovitz-Eldor, M.D., D.Sc.,1,
3,
5 and Ofer Binah, Ph.D.1,
2,
3
*
1The Sohnis Family Stem Cells Center, Technion - Israel Institute of Technology, Haifa, Israel;
2The Rappaport Family Institute for Research in the Medical Sciences, Technion - Israel Institute of Technology, Haifa, Israel;
3Ruth & Bruce Rappaport Faculty of Medicine, Technion - Israel Institute of Technology, Haifa, Israel;
4Department of Pediatric Cardiology, Rambam Health Care Campus, Haifa, Israel; and
5Department of Obstetrics and Gynecology, Rambam Health Care Campus, Haifa, Israel
This is an open-access article. All its content, except where otherwise noted, is distributed under the terms of the Creative Commons Attribution License (
http://creativecommons.org/licenses/by/3.0
), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
|
Catecholaminergic polymorphic ventricular tachycardia (CPVT) is an inherited arrhythmogenic cardiac disorder characterized by life-threatening arrhythmias induced by physical or emotional stress, in the absence structural heart abnormalities. The arrhythmias may cause syncope or degenerate into cardiac arrest and sudden death which usually occurs during childhood. Recent studies have shown that CPVT is caused by mutations in the cardiac ryanodine receptor type 2 (RyR2) or calsequestrin 2 (CASQ2) genes. Both proteins are key contributors to the intracellular Ca2+ handling process and play a pivotal role in Ca2+ release from the sarcoplasmic reticulum to the cytosol during systole. Although the molecular pathogenesis of CPVT is not entirely clear, it was suggested that the CPVT mutations promote excessive sarcoplasmic reticulum Ca2+ leak, which initiates delayed afterdepolarizations (DADs) and triggered arrhythmias in cardiac myocytes. The recent breakthrough discovery of induced pluripotent stem cells (iPSC) generated from somatic cells (e.g. fibroblasts, keratinocytes) now enables researches to investigate mutated cardiomyocytes generated from the patient’s iPSC. To this end, in the present article we review recent studies on CPVT iPSC-derived cardiomyocytes, thus demonstrating in the mutated cells catecholamine-induced DADs and triggered arrhythmias.
Keywords:
Adrenergic stimulation, cardiac myocytes, catecholaminergic polymorphic ventricular tachycardia (CPVT), delayed afterdepolarizations, induced pluripotent stem cells (iPSC), inherited arrhythmias
|
Sudden cardiac death caused by a ventricular arrhythmia is a disastrous event, especially when it occurs in young individuals. Among the five major arrhythmogenic disorders occurring in the absence of structural heart diseases is catecholaminergic polymorphic ventricular tachycardia (CPVT), which is a highly lethal form of inherited arrhythmogenic disease characterized by adrenergically mediated polymorphic ventricular tachycardia.
1
–
4
In response to physical activity or emotional stress, this disease is characterized by episodes of syncope, seizures, or sudden death.
1
,
2
CPVT was first described as a case report by Reid et al.
5
who reported on a bidirectional ventricular tachycardia triggered by physical effort and emotional stress in a 6-year-old girl (who survived a cardiac arrest) with no evidence of any structural heart disease. Later on, in 1978 and 1995, Coumel et al.
6
and Leenhardt et al.
7
reported a series of cases in familial as well as in sporadic forms of the arrhythmia and introduced the term catecholaminergic polymorphic ventricular tachycardia (CPVT) to refer to a disease characterized by adrenergically mediated bidirectional and/or polymorphic ventricular tachycardia in the absence of cardiac pathology. In 2001, Priori et al.
8
and Lahat et al.
9
identified mutations in the cardiac ryanodine receptor (RyR2) and cardiac calsequestrin (CASQ2) genes, respectively, underlying autosomal dominant and autosomal recessive forms of the disease. RyR2 is a cardiac Ca2+ release channel located on the sarcoplasmic reticulum (SR) membrane and has a key role in the process of calcium-induced calcium release (CICR) during the excitation–contraction (E–C) coupling.
10
,
11
CASQ2 is a high-capacity, low-affinity Ca2+-binding protein, operating as a major Ca2+-buffering factor, and together with RyR2 forms the SR Ca2+ release unit.
8
,
10
,
11
Since both proteins are key players in the E–C coupling process, the functional derangements in intracellular Ca2+-handling resulting from the mutated RyR2 and CASQ2 genes may cause delayed afterdepolarizations (DADs), which constitute the major electrophysiological mechanism underlying CPVT.
2
,
12
The prevalence of CPVT in the population is not completely known and has been estimated as 1:10,000.
4
If left untreated, 80% of CPVT patients will develop symptoms (ventricular tachycardia, ventricular fibrillation, syncope, sudden death) by the age of 40, with overall mortality of 30%–50%.
13
Because β-adrenergic blockers and implantable cardioverter defibrillator (ICD) therapy can rescue most CPVT cases, early diagnosis by means of clinical evaluations and genetic screening is possible and crucial. Hence, ICD may be considered for primary prevention of a cardiac arrest in CPVT patients in whom severe ventricular arrhythmias or recurrent syncope are observed in the presence of β-adrenergic blocking therapy.
2
In general, two genetic variants of CPVT have been identified. One is transmitted as an autosomal dominant trait caused by mutations in the gene encoding RyR2 (CPVT1) which is responsible for 50%–55% of all CPVT patients. Presently, more than 150 mutations have been identified in the RyR2 gene,
14
preferentially located in four highly conserved regions (domains I–IV) of the gene.
15
The second variant is an autosomal recessive form caused by mutations in the cardiac specific isoform of the calsequestrin gene CASQ2 (CPVT2) which represents only 3%–5% of CPVT patients.
4
To date, 15 CASQ2 mutations have been identified in the short arm of chromosome 1 which lead to severe decrease or complete loss of the CASQ2 protein.
4
|
THE MOLECULAR MECHANISM UNDERLYING CPVT
Key Elements of the Excitation–Contraction Coupling Machinery The delicate balance that regulates Ca 2+ fluxes between the intracellular compartment and the extracellular space in cardiomyocytes is critical to ensure cellular viability, preserve normal contractile function, and to provide a stable heart rhythm.
2
,
16
During the plateau phase of the cardiac action potential, a small amount of Ca 2+ enters the cardiomyocytes through the voltage-dependent L-type Ca 2+ channels, causing Ca 2+ release into the cytosol through the RyR2 channel located in the SR membrane. This process of CICR is the basis of cardiac E–C coupling.
11
,
16
,
17
The attainment of higher concentrations of cytosolic Ca 2+ causes activation of the contractile filaments of the cardiac sarcomere, which is followed by diminution of Ca 2+ concentration to the diastolic level, thus causing relaxation. Ca 2+ levels are lowered to diastolic values by means of three systems: 1) the sarcoplasmic endoplasmic reticulum calcium (SERCA2, the cardiac SERCA) which is responsible for reuptake of ∼75% of Ca 2+ into the SR; 2) The sodium–calcium exchanger (NCX) which extrudes the remaining portion of cytosolic Ca 2+ from the cytoplasm; and 3) a small portion of the Ca 2+ is extruded to the extracellular space by means of an ATP-operated Ca 2+ pump.
11
The Ca2+ Release Unit (CRU) RyR2 is a large homotetrameric Ca 2+ release channel located on the SR membrane. The RyR2 channels are composed of four pore-forming monomers, comprising a relatively small C-terminal transmembrane domain and a large N-terminal domain that protrudes into the cytosol. The cytoplasmic domain of RyR2 is stabilized by FKBP12.6 and is essential for channel closure during diastole.
8
,
17
,
18
The CASQ2 is the major Ca 2+ storage protein in the SR and is capable of binding luminal Ca 2+ (40–50 Ca 2+ ions/molecule) during diastole in order to prevent Ca 2+ precipitation and to reduce the ionic Ca 2+ concentration.
19
On the luminal side, RyR2 binds junctin and triadin, which anchor the Ca 2+-buffering protein CASQ2,
11
collectively forming the SR Ca 2+ release unit (CRU). The CRU is responsible for SR Ca 2+ release, which is triggered by increased cytosolic Ca 2+ resulting from opening of the L-type channel (CICR).
11
In addition, CASQ2 has been suggested to modulate the activity of RyR2 directly.
20
Under adrenergic stimulation, β-adrenergic receptors activate a GTP-binding protein that stimulates adenylyl cyclase to produce cAMP, which in turn activates protein kinase A (PKA). This kinase phosphorylates RyR2 and other central proteins related to E–C coupling, such as phospholamban and the L-type Ca 2+ channels, thus causing gain of function of Ca 2+ cycling in cardiomyocytes in response to adrenergic activation. FKBP12.6 stabilizes RyR2 in the closed state, and the hyperphosphorylation of RyR2 by PKA causes FKBP12.6 dissociation from RyR2, thereby increasing the open probability of RyR2.
21
Moreover, adrenergic stimulation also increases the activity of the SERCA pump via the phosphorylation of phospholamban by PKA which stabilizes SERCA.
The mechanism of CPVT
In-vitro studies suggested that the RyR2 and CASQ2 mutations cause the CRU to open spontaneously without being triggered by voltage-gated Ca 2+ influx, thereby leading to intracellular Ca 2+ overload.
1
,
2
Increased intracellular Ca 2+ can trigger early or delayed afterdepolarizations (oscillations of the membrane potential that occur during the plateau/ repolarization phase of the action potential or after its completion, respectively) that can reach the threshold potential and cause triggered activity.
15
Intracellular Ca 2+ overload leads to NCX activation which extrudes Ca 2+ in exchange for Na + with a stoichiometry of 1:3, thereby generating a net inward current (the so-called transient inward current, I Ti).
2
The transient inward current induces DADs which may reach threshold and trigger premature ventricular beats and ventricular arrhythmias (demonstrated in Figure 1) by a mechanism called triggered activity.
2
|
Figure 1
Ca2+-induced Ca2+ release (CICR), store overload-induced Ca2+ release (SOICR), and triggered arrhythmia.
|
|
EXPERIMENTAL MODELS OF CPVT
The electrophysiological mechanisms of CPVT, which result from mutations in the RyR2 and CASQ2 genes, were investigated in several in-vitro and in-vivo models.
12
,
22
–
24
The first CPVT transgenic mouse model was produced by Priori’s group in 2005. These authors introduced the RyR2 R4496C mutation into the mouse genome by homologous recombination and successfully reproduced the human phenotype.
24
This in-vivo model proved the concept that RyR2 mutations may cause polymorphic and bidirectional ventricular tachycardia in a knock-in mouse model and mimic the clinical phenotype of CPVT patients.
24
Subsequently, Priori’s group demonstrated that myocytes isolated from the heart of the RyR2 R4496C knock-in mouse generated DADs and triggered activity upon exposure to β-adrenergic stimulation, while the control myocytes isolated from wild-type mouse did not.
25
Similar results were reported by Kannankeril et al. who generated a knock-in mouse model of RyR2 R176Q mutation and demonstrated the disease phenotype in vivo and in vitro.
26
Priori’s group was also the first to generate in-vitro modeling of the CASQ2 D307H mutation using infected rat myocytes with adenoviruses engineered to express the mutated CASQ2. In this study the authors demonstrated that the mutated myocytes developed DADs upon adrenergic stimulation while the control myocytes did not, as shown in Figure 2. This model provided the first evidence that mutant CASQ2 can generate DADs.
12
Using the same model of rat myocytes infected with adenoviruses, Terentyev et al. and di Barletta el al. generated in-vitro models of the CASQ2 R33Q and L167H mutations, respectively, and reported on the generation of DADs in the mutated myocytes.
22
,
23
Nonetheless, these models suffer from several limitations because the myocytes express both the mutant and the endogenous wild-type CASQ2. In 2006, Knollmann et al. generated CASQ2-null (CASQ2−/−) mice which were viable and exhibited the human-like arrhythmias. Despite the absence of CASQ2, these animals maintained relatively normal Ca2+ release and contractile function. However, myocytes isolated from these mice had exceptional increases in SR volume, increased gain of Ca2+-induced SR Ca2+ release, and increased diastolic SR Ca2+ leak.
27
Other transgenic models with deficient RyR2 or CASQ2 genes were generated and have confirmed the correlation between the arrhythmogenic phenotype to the mutant genes, although the observed phenotype did not always resemble the human disease.
28

|
Figure 2
Disturbances in rhythmic [Ca2+]i transients and membrane potential induced by ISO in myocytes expressing CASQ2D307H.
|
|
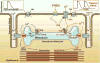
The recent discovery of genomic reprogramming of human somatic cells into induced pluripotent stem cells (iPSC) offers an innovative and practical approach to study inherited diseases including heart pathologies. Induced pluripotent stem cells are the product of somatic cell reprogramming to an embryonic-like state. Human iPSC have been generated from various types of somatic cells, most commonly skin fibroblasts, but also from hair keratinocytes isolated from plucked human hair which offers significant advantages over isolating skin fibroblasts through an invasive surgical procedure.
29
This process of reprogramming occurs by the introduction of a defined and limited set of transcription factors and by culturing these cells under embryonic stem cell conditions. Somatic cell reprogramming by the induction of four ectopic genes (OCT3/4, SOX2, C-MYC, and KLF4) by retroviral insertion was first described in mouse cells,
30
and later on in human cells,
31
by Yamanaka’s group. This new technology allows us to investigate cardiac disorders in vitro and opens new opportunities for investigating the diseases’ mechanism in vitro, developing new drugs, predicting their toxicity, and optimizing current treatment strategies (Figure 3). Recently, several groups reported on the generation of patient-specific iPSC of the inherited arrhythmias—LEOPARD syndrome,
32
long-QT1,
33
long-QT2,
34
,
35
and Timothy syndrome
36
—and demonstrated the capacity of these cells to give rise to functional cardiomyocytes that display the electrophysiological characteristics of the disorder. In the last year, three groups reported on the generation of iPSC-derived cardiomyocytes from CPVT2
37
and CPVT1
38
,
39
patients. In this regard, our group was the first to generate cardiomyocytes from CPVT patients carrying the missense D307H in the CASQ2 gene, which in response to β-adrenergic stimulation generated DADs and triggered activity.
37
Specifically, Novak et al. reported on the generation of cardiomyocytes from two CPVT2 patients: a 12-year-old boy and a 30-year-old woman carrying the missense mutation D307H in the CASQ2 gene, which exhibited the key features of catecholaminergic-mediated arrhythmogenesis.
37
The D307H mutation (associated with a change from a negatively charged aspartic acid to a positively charged histidine) causes reduced affinity of the mutant CASQ2 to Ca2+, thereby resulting in Ca2+ spillover during adrenergic stress.
4
To decipher the cellular mechanisms of CPVT we performed patch clamp and intracellular Ca2+ and contraction measurements from iPSC cardiomyocytes generated from healthy and diseased individuals. In agreement with previous studies reporting a lower resting heart rate among CPVT patients,
40
,
41
we found that the mean spontaneous beating rate of CPVT iPSC cardiomyocytes was slower by 34% than control cardiomyocytes. These findings still need to be established in a larger number of cells from different clones. The adrenergically mediated arrhythmogenic features of CPVT2 cardiomyocytes were demonstrated by exposing the cells to the β-adrenergic agonist isoproterenol. While isoproterenol or pacing were not arrhythmogenic in control cardiomyocytes, in 47% of the CPVT cardiomyocytes isoproterenol caused the generation of after-contractions (which are the mechanical equivalent of DADs) and triggered beats (Figure 4B and C), and in 33% of mutated cardiomyocytes pacing alone caused after-contractions in the absence of isoproterenol. Further, in contrast to control cardiomyocytes in which isoproterenol commonly causes positive inotropic and lusitropic (increased rate of relaxation) effects, CPVT cardiomyocytes were unresponsive. Further, in CPVT (but not in control) cardiomyocytes, isoproterenol caused marked elevation in the resting tension level, probably resulting from a prominent diastolic [Ca2+]i rise (Figure 4E). This last-mentioned phenomenon is the principal feature of the CASQ2 mutation, as previously demonstrated in paced cardiomyocytes derived from a CASQ2-deficient mutant mouse.
42
Next, by means of the patch clamp technique, we
37
demonstrated (Figure 5) for the first time that, in addition to the familiar DADs occurring in CPVT2-mutant cardiomyocytes in response to β-adrenergic stimulation,
8
,
12
,
43
isoproterenol also caused arrhythmogenic depolarizing oscillatory prepotentials, which were originally described in cardiac muscle by Bozler in 1942.
44
,
45
In contrast to DADs which follow the action potential and therefore appear during the early diastolic depolarization, oscillatory prepotentials are defined as diastolic voltage oscillations which appear during the late diastolic depolarization.
46
,
47
The oscillatory prepotentials have longer duration than DADs, they overshoot and undershoot the late diastolic depolarization, and they grow progressively in amplitude until reaching the threshold and initiating spontaneous activity.
46
,
47
Further, 75% of the CPVT cardiomyocytes exposed to isoproterenol developed DADs (Figure 5B), oscillatory prepotentials (Figure 5C), or both (Figure 5D). Finally, ultrastructural analysis of CPVT2 cardiomyocytes showed a small expansion of the SR cisternae,
37
as previously reported in a mouse model of CPVT with a deficient cardiac CASQ2.
27
,
43
,
48
This expansion was suggested to constitute a compensatory response for the loss of SR Ca2+ buffering by the mutated CASQ2.
27

|
Figure 3
Induced pluripotent stem cells—derivation and applications.
|

|
Figure 4
The effects of isoproterenol on the [Ca2+]i transients and contractions in control and CPVT iPSC cardiomyocytes.
|

|
Figure 5
DADs and oscillatory prepotentials in CPVT iPSC cardiomyocytes induced by isoproterenol.
|
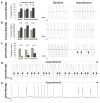
More recently, Fatima and co-workers generated CPVT1-specific iPSC from a 46-year-old woman diagnosed with CPVT1, who carried the novel heterozygous autosomal dominant missense mutation F2483I in the RYR2 gene. This mutation is localized in the FKBP12.6-binding domain of the RYR2 protein.
38
In agreement with our findings,
37
these authors showed that while in all control cardiomyocytes isoproterenol caused a positive chronotropic effect, but no arrhythmias, in 57.9% of the CPVT cardiomyocytes isoproterenol caused a negative chronotropic effect. Further, in 34.2% of the RyR2-mutant cells isoproterenol caused putative DADs and arrhythmias (Figure 6). Finally, using [Ca2+]i imaging Fatima et al. showed that CPVT1 cardiomyocytes exhibit higher amplitudes and longer durations of spontaneous local Ca2+ release events already at basal state compared to the control cardiomyocytes. These findings are consistent with the irregular SR Ca2+ release observed in cells expressing the mutant RyR2 that underlie CPVT1.
25
|
Figure 6
Electrophysiological characterization of control and CPVT1 cardiomyocytes (CM).
|
In a recent study Jung et al. reported on the generation of iPSC from a 24-year-old CPVT1 woman carrying the novel RyR2 S406L mutation.
39
The S406L mutation is located in the N-terminal domain of the RyR2 channel, which is one of the three hot spots for CPVT-associated RyR2 mutations. Based on immunofluorescence staining of control and CPVT cardiomyocytes, the authors proposed that the S406L mutation does not interfere with trafficking of the homotetrameric channel. These authors further demonstrated that increasing the stimulation frequency was associated with a higher percentage of cells with abnormal Ca2+ handling in CPVT than in control cells. This frequency-induced stress is compatible with our findings
37
that stimulation alone caused arrhythmias in CPVT but not in control cardiomyocytes. Jung et al. also reported that control and CPVT1 cardiomyocytes had similar diastolic and systolic Ca2+ levels, as well as comparable SR Ca2+ content, determined by caffeine application. However, in the presence of isoproterenol the diastolic Ca2+ level was significantly elevated in CPVT1 cardiomyocytes compared to control cells, while systolic Ca2+ remained similar; these findings are in agreement with the effects of isoproterenol on CPVT2 cardiomyocytes.
37
Moreover, in contrast to control cardiomyocytes, SR Ca2+ load was not increased by isoproterenol in CPVT1 cells. Furthermore, under basal conditions, CPVT cardiomyocytes displayed abnormal Ca2+ sparks with a higher amplitude, prolonged (compared to control) plateau phase, longer duration at 50% peak amplitude, and longer decay time. In response to isoproterenol, in CPVT cardiomyocytes Ca2+ spark frequency increased compared to control cells, and the sparks had longer decay time. Finally, Jung et al. showed that in all CPVT1 cardiomyocytes stimulated with isoproterenol the DADs and triggered arrhythmias were abolished by the ryanodine antagonist dantrolene, suggesting that a defective inter-domain interaction within the RyR2 is the underlying arrhythmogenic mechanism of the S406L mutation.
|
CPVT is a complex disease which poses several challenges in the management of affected patients.
49
Despite the recent advancement in understanding the diverse aspects of CPVT, this fatal disease still presents high mortality rates among young and older individuals, and therefore there is an emerging need for developing targeted pharmacological agents. Patient-specific iPSC can provide useful platforms for the discovery of unprecedented insights into disease mechanisms, as well as new drugs.
50
Indeed, since the seminal breakthrough of generating iPSC,
31
,
51
several patient-specific iPSC lines were developed for a variety of cardiovascular syndromes
33
–
39
which demonstrated that their derived cardiomyocytes exhibit the electrophysiological features of the disorder, thus supporting the suitability of iPSC-derived cardiomyocytes to serve as a platform for exploring disease mechanisms in human genetic cardiac disorders. Hence, the derivation of cardiomyocytes from CPVT patients can provide the means to study, in the mutated myocytes, the functional changes and the underlying molecular mechanisms of CPVT, screen and develop candidate drugs on a patient-specific level, and thus advance our understanding of the disease and consequently improve its future clinical outcome.
Although DADs were described in vitro and in vivo in CPVT mouse models, the demonstration that these phenomena were responsible for arrhythmogenesis in humans was largely a consequence of genetic research.
52
Therefore, our findings
37
and those of others
38
,
39
demonstrating the generation of DADs and triggered arrhythmias in human CPVT patient-derived cardiomyocytes are of great importance. Finally, investigating the responsiveness, to anti-arrhythmic drugs, of CPVT-mutated cardiomyocytes from individual patients may give rise to the future application of “personalized medicine,” which is likely to reduce the morbidity and mortality of patients affected by inherited arrhythmias.
|
| CASQ2 |
cardiac calsequestrin |
| CICR |
calcium-induced calcium release |
| CPVT |
catecholaminergic polymorphic ventricular tachycardia |
| CRU |
calcium release unit |
| DADs |
delayed afterdepolarizations |
| EB |
embryoid body |
| E–C |
excitation–contraction |
| ICD |
intraventricular cardioverter defibrillator |
| iPSC |
induced pluripotent stem cells |
| RyR2 |
cardiac ryanodine receptor |
| SR |
sarcoplasmic reticulum |
|
|
|
5.
Reid DS, Tynan M, Braidwood L, Fitzgerald GR. Bidirectional tachycardia in a child. A study using His bundle electrography.
Br Heart J. 1975;37:339–44.
http://dx.doi.org/10.1136/hrt.37.3.339
.
6.
Coumel P, Fidelle J, Lucet V, Attuel P, Bouvrain Y. Catecholaminergic-induced severe ventricular arrhythmias with Adams-Stokes syndrome in children: report of four cases.
Br Heart J. 1978;40:28–38.
7.
Leenhardt A, Lucet V, Denjoy I, Grau F, Ngoc DD, Coumel P. Catecholaminergic polymorphic ventricular tachycardia in children. A 7-year follow-up of 21 patients.
Circulation. 1995;915:1512–9.
http://dx.doi.org/10.1161/01.CIR.91.5.1512
.
8.
Priori SG, Napolitano C, Tiso N, et al. Mutations in the cardiac ryanodine receptor gene (hRyR2) underlie catecholaminergic polymorphic ventricular tachycardia.
Circulation. 2001;103:196–200.
http://dx.doi.org/10.1161/01.CIR.103.2.196
.
9.
Lahat H, Pras E, Olender T, et al. A missense mutation in a highly conserved region of CASQ2 is associated with autosomal recessive catecholamine-induced polymorphic ventricular tachycardia in Bedouin families from Israel.
Am J Hum Genet. 2001;69:1378–84.
http://dx.doi.org/10.1086/324565
.
10.
Fabiato A. Calcium-induced release of calcium from the cardiac sarcoplasmic reticulum.
Am J Physiol. 1983;245:C1–14.
13.
Cerone MCB, Boise R, Memmi M, et al. Clinical and molecular characterization of a large cohort of patients affected with catecholaminergic polymorphic ventricular tachycardia (abstract).
Circulation. 2004;110(Suppl II):552.
16.
Chelu MG, Wehrens XH. Sarcoplasmic reticulum calcium leak and cardiac arrhythmias.
Biochem Soc Trans. 2007;35(Pt 5):952–6.
18.
Liu N, Priori SG. Disruption of calcium homeostasis and arrhythmogenesis induced by mutations in the cardiac ryanodine receptor and calsequestrin.
Cardiovasc Res. 2008;77:293–301.
http://dx.doi.org/10.1093/cvr/cvm004
.
22.
Terentyev D, Nori A, Santoro M, et al. Abnormal interactions of calsequestrin with the ryanodine receptor calcium release channel complex linked to exercise-induced sudden cardiac death.
Circ Res. 2006;98:1151–8.
http://dx.doi.org/10.1161/01.RES.0000220647.93982.08
.
23.
di Barletta MR, Viatchenko-Karpinski S, Nori A, et al. Clinical phenotype and functional characterization of CASQ2 mutations associated with catecholaminergic polymorphic ventricular tachycardia.
Circulation. 2006;114:1012–9.
http://dx.doi.org/10.1161/CIRCULATIONAHA.106.623793
.
24.
Cerrone M, Colombi B, Santoro M, et al. Bidirectional ventricular tachycardia and fibrillation elicited in a knock-in mouse model carrier of a mutation in the cardiac ryanodine receptor.
Circ Res. 2005;96:e77–82.
http://dx.doi.org/10.1161/01.RES.0000169067.51055.72
.
26.
Kannankeril PJ, Mitchell BM, Goonasekera SA, et al. Mice with the R176Q cardiac ryanodine receptor mutation exhibit catecholamine-induced ventricular tachycardia and cardiomyopathy.
Proc Natl Acad Sci U S A. 2006;103:12179–84.
http://dx.doi.org/10.1073/pnas.0600268103
.
27.
Knollmann BC, Chopra N, Hlaing T, et al. Casq2 deletion causes sarcoplasmic reticulum volume increase, premature Ca2+ release, and catecholaminergic polymorphic ventricular tachycardia.
J Clin Invest. 2006;116:2510–20.
28.
Cerrone M, Napolitano C, Priori SG. Catecholaminergic polymorphic ventricular tachycardia: A paradigm to understand mechanisms of arrhythmias associated to impaired Ca2+ regulation.
Heart Rhythm. 2009;6:1652–9.
http://dx.doi.org/10.1016/j.hrthm.2009.06.033
.
29.
Novak A, Shtrichman R, Germanguz I, et al. Enhanced reprogramming and cardiac differentiation of human keratinocytes derived from plucked hair follicles, using a single excisable lentivirus.
Cell Reprogram. 2010;12:665–78.
http://dx.doi.org/10.1089/cell.2010.0027
.
32.
Carvajal-Vergara X, Sevilla A, D’Souza SL, et al. Patient-specific induced pluripotent stem-cell-derived models of LEOPARD syndrome.
Nature. 2010;465:808–12.
http://dx.doi.org/10.1038/nature09005
.
34.
Matsa E, Rajamohan D, Dick E, et al. Drug evaluation in cardiomyocytes derived from human induced pluripotent stem cells carrying a long QT syndrome type 2 mutation.
Eur Heart J. 2010;32:952–62.
http://dx.doi.org/10.1093/eurheartj/ehr073
.
36.
Yazawa M, Hsueh B, Jia X, et al. Using induced pluripotent stem cells to investigate cardiac phenoltypes in Timothy syndrome.
Nature. 2011;471:230–4.
http://dx.doi.org/10.1038/nature09855
.
38.
Fatima A, Xu G, Shao K, et al. In vitro modeling of ryanodine receptor 2 dysfunction using human induced pluripotent stem cells.
Cell Physiol Biochem. 2011;28:579–92.
http://dx.doi.org/10.1159/000335753
.
39.
Jung CB, Moretti A, Mederos y Schnitzler M, et al. Dantrolene rescues arrhythmogenic RYR2 defect in a patient-specific stem cell model of catecholaminergic polymorphic ventricular tachycardia.
EMBO Mol Med. 2012;4:180–91.
http://dx.doi.org/10.1002/emmm.201100194
.
40.
Postma AV, Denjoy I, Kamblock J, et al. Catecholaminergic polymorphic ventricular tachycardia: RYR2 mutations, bradycardia, and follow up of the patients.
J Med Genet. 2005;42:863–70.
http://dx.doi.org/10.1136/jmg.2004.028993
.
42.
Song L, Alcalai R, Arad M, et al. Calsequestrin 2 (CASQ2) mutations increase expression of calreticulin and ryanodine receptors, causing catecholaminergic polymorphic ventricular tachycardia.
J Clin Invest. 2007;117:1814–23.
http://dx.doi.org/10.1172/JCI31080
.
43.
Dirksen WP, Lacombe VA, Chi M, et al. A mutation in calsequestrin, CASQ2D307H, impairs sarcoplasmic reticulum Ca2+ handling and causes complex ventricular arrhythmias in mice.
Cardiovasc Res. 2007;75:69–78.
http://dx.doi.org/10.1016/j.cardiores.2007.03.002
.
44.
Bozler E. The initiation of impulses in cardiac muscle.
Am J Physiol. 1942;138:273–82.
46.
Kim EM, Choy Y, Vassalle M. Mechanisms of suppression and initiation of pacemaker activity in guinea-pig sino-atrial node superfused in high [K+]o.
J Mol Cell Cardiol. 1997;29:1433–45.
http://dx.doi.org/10.1006/jmcc.1997.0382
.
48.
Rizzi N, Liu N, Napolitano C, et al. Unexpected structural and functional consequences of the R33Q homozygous mutation in cardiac calsequestrin: a complex arrhythmogenic cascade in a knock in mouse model.
Circ Res. 2008;103:298–306.
http://dx.doi.org/10.1161/CIRCRESAHA.108.171660
.
50.
Kiskinis E, Eggan K. Progress toward the clinical application of patient-specific pluripotent stem cells.
J Clin Invest. 2010;120:51–9.
http://dx.doi.org/10.1172/JCI40553
.
|
