Potential Health Risks
As new biotechnologies enter clinical trials, one needs to address the halachic issues related to someone who wants to volunteer for any phase I clinical trial and in particular MRT. One of the main concerns in volunteering in a clinical trial relates to the halacha that it is forbidden for a Jew to place his or her life or health in an unreasonably dangerous situation.
In addition, the Rambam states that a person must live a lifestyle that preserves health, and the Talmud and the Codes of Halacha mention a wide variety of other examples of activities that promote well-being and curtail health risks. These include:
-
eating healthily and not becoming overweight,
-
not putting one’s mouth directly onto a pipe in order to drink water,
-
not drinking water drawn from a river at night when one is unable to inspect for parasites or drink liquids that have been left exposed and unattended where there is a possibility, albeit remote, that a snake may have deposited venom,
-
not eating food that might be tainted or poisoned etc.,
-
not engaging in any activity that causes physical wounds or injuries to oneself.16–18
Endangering one’s own life is not uniformly prohibited. There is a Biblical duty to rescue someone from danger, such as rescuing someone from drowning or from a wild animal or from robbers.19,20 Moreover, we are Biblically commanded, “Do not stand by idly over your friend’s blood” (Leviticus 19:16). An underlying question in this halacha is “what degree of danger can a person incur to save another life?” The Netziv in He’emek Sh’ailah #129 states that a person cannot place him/herself in serious danger even to save another person. However, one is permitted to accept a modicum of danger as an act of beneficence (middas chassidus). However, there must be careful consultations with physicians and halachic ethicists to determine the modicum of danger that is allowed for each specific case. (For more information considering risks and dangers related to health care situations, see Immanuel Jakobovits.21)
The major health risk to women volunteering for MRT is related to ovarian hormonal stimulation to retrieve their eggs. Since most women do not present with serious side effects resulting from ovarian hormonal stimulation, volunteering for a clinical trial as a mitochondrial donor even though they may not directly benefit would certainly be considered an act of beneficence, a great chesed, and is permitted. Similarly, it is a great chesed for women with mitochondrial mutations to volunteer in MRT trials in order to have healthy children. In volunteering for such trials, these women enter into the realm of a potentially real reward—the birth of a genetically related healthy child.
What about risking health for the financial gains gleaned when healthy individuals enroll in a clinical trial? This issue also is relevant when choosing a career. Is there a difference when a person chooses a career with health risks (such as being a fireman or policeman) and volunteering to enroll in a clinical trial? We maintain that, in both situations, financial gain provides a valid halachic reason either to choose a dangerous career or to volunteer to enroll in a clinical trial. One is also halachically permitted to enter into a clinical trial for financial reasons. The risks to one’s health for financial gains are no different than choosing a career that involves an increment of risk and danger. Financial gains from a chosen employment such as building bridges, constructing underground tunnels, or physicians choosing to specialize in infectious diseases are all halachically permitted, despite the associated dangers of these professions.22,23
Since halacha provides a healthy person with the autonomy to volunteer for a clinical trial, individuals who present with medical conditions where no alternative therapies exist may surely volunteer to engage in new clinical trials, even when there may be a great medical risk. Thus, individuals with valid health or financial considerations would be permitted to volunteer in a clinical trial even when there may be a risk of potential harm. Since the health risks of MRT are not fully understood, halacha provides infertile women the autonomy to engage in these technologies in order to have healthy children. However, these women must fully understand the risks and benefits of this technology.
Halachic Reasons to Participate in Clinical Trials
There are both positive and negative halachic reasons to volunteer for clinical trials. The Midrash states “Before the Torah was given, the Creator required that people contribute to the formation of a viable society.”
24 This is a pre-Biblical commandment. God did not create a world to be empty but to be populated.
25 However, in order to fulfill the will of God to populate a functioning world, society must establish water supplies and sewer systems as part of the mitzvah to create a functioning society. This theme is reflected in the Midrash’s understanding of Tehilim (Psalms 50:21) “He who offers a
korban todah [a thanksgiving offering] brings honor to God.” The Midrash provides four examples of what is considered an offering of thanks: (1) people who remove obstacles from roads to allow fluid travel, (2) people who light candles in public so that others can travel safely at night, (3) merchants who make the effort to tithe their produce before selling to the public, and (4) people who agree to become teachers of young children—a career that is quite challenging and difficult. None of these examples mentioned in the Midrash are classical ritual actions but are activities that reflect social responsibilities.
Moreover, the Torah teaches the virtues of contributing to the general society in relating two stories of our forefathers, Avraham and Yaakov. When Avraham returned to the Land of Canaan from Egypt the Talmud states that he would stay overnight in the same lodgings that he stayed in when he first went down to Egypt.26,27 The lesson here is to “teach the proper way of conduct” that changing lodgings might be viewed as dissatisfaction with the hotel in which Avraham stayed when he first went to Egypt. In order for hotels to be successful they require a loyal clientele. It is therefore our social responsibility that we support good hotels to ensure a viable system of travel. The second example is related to Yaakov when he came to Shechem and the verse states, “he encamped before the city.”28, The Talmud states that Yaakov engaged in social responsibility to the members of Shechem by teaching them how to: (1) organize a currency system, (2) establish a market place to sell goods, and (3) build bath houses.29 Yaakov did not teach the people of Shechem ritual observances but rather social systems to benefit the municipality. These lessons that we learned from our forefathers highlight how engaging in social responsibility, such as volunteering in MRT studies, brings honor to God. In addition, there is a real need for Jews to develop friendly relations with members of other faiths in a world where so many ethnic groups live together.
The negative halachic aspect of volunteering for clinical trial may fall under the prohibition of aiva (anti-Jewish enmity). Failure to participate in social responsibility can incur the disdain of society. This disdain can also incur anger by members of the society that could lead to jeopardizing the health care or even lives of Jews. The halachic ruling that a Jewish physician should transgress the shabbath in treating members of other faiths is based upon the concern that failure to treat non-Jews by Jewish physicians could have severe repercussions and might even endanger Jews in the community.30
One example of the consequences of aiva is organ transplantation. Since a significant percentage of orthodox Jews believe that death is defined by cessation of heart beats, they cannot serve medically as heart donors. In many countries, anyone who is not an organ donor has a lower priority in receiving organ transplants. Living American organ donors, for example, who later need kidney transplants have much shorter waiting times, and they receive higher-quality kidneys compared with similar people on the waiting list who did not designate themselves as organ donors.31 In countries such as Israel and Singapore, registered organ donors are given priority on organ waiting lists. This provides an incentive for organ donor registration and has the potential to increase the pool of deceased donor organs.32 Moreover, as a consequence of halachic scholars not accepting brain stem death, the refusal to donate organs makes it difficult for Israelis to receive critical organs.33
As stated above, volunteering for clinical trials represents an act of beneficence (chesed) and encompasses a moral obligation of a Jew to contribute to improving the health care of a society and thus bring honor to our God. Thus, encouraging Jews to volunteer for MRT trials is quite important in a society where Jews and non-Jews interact in almost every aspect of life. In addition, volunteering for such trials would prevent enmity and promote good will. While the number of Jews who would volunteer for any phase I clinical trials is far fewer than those considering organ donation, the current atmosphere in the United States and other Western countries views orthodox Jews under a microscope for impropriety. Aiva is more than merely the avoidance of actions that may lead to enmity but encompasses preventative actions as well. The act of orthodox Jews participating in a socially appropriate activity adds kavod and pride to our people and mitigates aiva. In the case of MRT, there are no real medical alternatives that can produce a genetically related healthy child from a woman with mitochondrial mutations. Volunteering to be an egg donor in this case, even for financial reasons, would be encouraged and halachically permitted.
The verse that is quoted in birchat hamazon
(“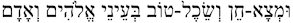 ” [“So shalt thou find grace and good favor in the sight of God and man”]) alludes to the theme that Jews should engage in activities that promote good will and social responsibility.34 The verse suggests that the actions an individual performs should be those that find grace and sound judgment in the eyes of God and humankind. Here the term adam represents universal human beings, not just members of B’nei Israel. Thus, in this bracha, we ask God to help us act in ways that achieve the approval of God and society.
” [“So shalt thou find grace and good favor in the sight of God and man”]) alludes to the theme that Jews should engage in activities that promote good will and social responsibility.34 The verse suggests that the actions an individual performs should be those that find grace and sound judgment in the eyes of God and humankind. Here the term adam represents universal human beings, not just members of B’nei Israel. Thus, in this bracha, we ask God to help us act in ways that achieve the approval of God and society.
Halachic Issues Related to the Newborn Child
There are, of course, halachic considerations concerning the newborn that emerge from such mitochondrial replacement therapies. First, how does halacha view the fact that the child was created using three genetic donors? This issue has been discussed in greater detail in a previous publication regarding human cloning, claiming that all three genetic donors may have some halachic status.
35,36 One might, however, argue that MRT differs from traditional cloning in that the mitochondrial donor is only supplying an extremely small amount of DNA (37 genes out the over 20,000 genes in the human genome) to the embryo. The response to this argument might hinge on the fact that the mitochondrial DNA is critical to life. An embryo cannot develop into a child without mitochondrial DNA, and this simply cannot be ignored despite the fact that mitochondrial DNA only contributes a fraction of the genetic information.
There are other complex issues related to MRT that must be evaluated in the future. What is the religious status of child, for example, when one of the female donors is not Jewish? In halacha, religion is maternally transmitted. If an embryo is conceived using DNA from two women, do both have to be Jewish for the child to be considered Jewish? Another example relates to pidyon haben. Normally, the first male child born from a mother who is a bat-Levi or bat-Kohen does not require a pidyon haben. What about a child born from a mother who is a bat-Kohen or bat-Levi but received its mitochondrial DNA from a donor who is a bat-Yisrael? Similarly, would a child born from a bat-Yisrael require a pidyon haben when the mitochondrial donor was a bat-Levi or bat-Kohen? A third halachic question is whether a MRT-generated child can marry a direct relative of the mitochondrial donor? Halacha has specific criteria permitting or prohibiting marriage to a relative. All these halachic issues are quite complex and will require halachic analyses beyond the scope of this article. We hope to address them in future articles (YY’H).

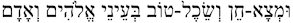 ” [“So shalt thou find grace and good favor in the sight of God and man”]) alludes to the theme that Jews should engage in activities that promote good will and social responsibility.
” [“So shalt thou find grace and good favor in the sight of God and man”]) alludes to the theme that Jews should engage in activities that promote good will and social responsibility.