HISTORY OF MODERN LIVER SURGERY
The German surgeon Carl Johann August Langenbuch performed the first successful hepatic resection in 1888 (he was the first to perform cholecystectomy in 1882).1 He resected a part of the left lobe of the liver after ligating the vascular pedicles. The pathologic examination of the specimen revealed normal liver.
The first steps leading to modern liver surgery began in the late 1950s. Scattered publications from the United States describing a limited series of liver resections that had met with some success were published. The technique of liver resection at that time was ill defined.2,3
Concomitantly, in 1952 in France Lortat Jacob published a manuscript on his experiences performing anatomical liver resections: right hepatectomy after vascular control of the right liver.4 In 1956, Claude Couinaud,5,6 after studying casts of the liver, published his book, The liver – anatomicalstudies and surgical studies. He showed that, according to the distribution of the portal blood, the liver contains four parts that are subdivided by the hepatic veins into eight segments. He coined the segment numbers as follows: one for the central segment and two through eight for the seven other segments in a clock-wise fashion. This segmental division of the liver is the basis of modern functional and surgical liver anatomy.
Despite the anatomical discoveries of the 1950s, their application in surgical practice was limited. No clinical methods existed that could detect the existence of small liver tumors that might have required segmental resections. Physicians made their diagnoses based on physical symptoms (pain or a palpable tumor), at which point it was usually already too late for surgery. Only large liver resections could be performed.
The introduction of ultrasound in the early 1980s7,8 into common clinical practice allowed the clinician to diagnose asymptomatic small liver tumors of 2 and 3 cm and paved the way to rapid development of liver surgery. In fact, modern liver surgery began when functional liver anatomy discovered 30 years earlier was applied, enabling segmental liver surgery.
The paper with the somewhat provocative title of “Surgical anatomy and anatomical surgery of the liver”9 published in 1982 was a turning-point in the practice of liver surgery. Navigating surgery on the basis of anatomy eliminated the use of “atypical” or “non-anatomical” resections of the liver which had resulted in bleeding, increased morbidity, and mortality sometimes as a result of liver necrosis. Liver resections based on anatomy gained popularity and evolved into bloodless surgery that removed independent segments or groups of two or more segments.
In 1984, intraoperative ultrasound (IOUS) was introduced into practice.10–14 The technique allowed the surgeon to understand liver vasculature and biliary duct anatomy and rendered the liver transparent (Figure 1). In fact, the introduction of IOUS into liver surgery had the same impact as the inclusion of intraoperative cholangiography (IOC) fifty years earlier in biliary surgery.15 By finding the liver’s lines of division with IOUS and looking for a tumor, the surgeon could establish the relations among the portal elements, the hepatic veins, the hepatic parenchyma, and the tumor, and then decide what kind of anatomical resection needed to be carried out – “Hepatectomie à la carte” for each individual patient.  | Figure 1 The transparent liver showing the main arteries (in red), portal vein (in white), hepatic veins (in blue), and main parts of the biliary system (in green). Liver segments are numbered, and the round ligament is designated in yellow. |
Control of bleeding during liver resection is a major challenge for the surgeon. It is particularly difficult in cirrhotic liver due to the fibrotic nature of the liver tissue. The indication, as well as the type of clamping, depends mainly on the size and location of the lesions to be resected, the quality of the liver parenchyma, the surgeon’s preferences, and unexpected operative events. In 1958 Lin introduced the finger fracture technique, which involves crushing of liver parenchyma by the surgeon’s fingers under inflow occlusion so as to isolate vessels and bile ducts for ligation.16 This technique was subsequently improved through the use of surgical instruments such as a small Kelly clamp for blunt dissection (clamp crushing or “Kellyclasia”).9,17,18
In many centers, ultrasonic dissection using the Cavitron Ultrasonic Surgical Aspirator (CUSA; Tyco Healthcare, Mansfield, MA, USA) has become the standard technique for liver parenchyma dissection. With this technology, the liver tissue is fragmented with ultrasonic energy and aspirated, thus exposing small vascular and ductal structures that can be ligated or clipped with titanium hemoclips.19 The water jet dissector employs a pressurized jet of water instead of ultrasonic energy, in order to fragment the liver parenchyma tissue and expose the vascular and ductal structures.20 However, this technique has not become as popular as ultrasonic dissection.
To reduce blood loss during liver resection, intermittent inflow occlusion by clamping of the portal triad (Pringle maneuver) is frequently used.21,22 However, there is a limit to how long the Pringle maneuver can be applied. Prolonged inflow occlusion (over 120 minutes) may have deleterious effects on liver functions.23 Hepatic inflow occlusion can be directed to one side or to a segment by clamping the Glissonian pedicle at the hilum or inside the liver parenchyma (Figure 2).9,24–29 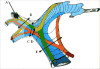 | Figure 2Glissonian pedicle elements: portal vein (in blue), hepatic artery (in red), and the bile ducts (in green). Hepatic inflow occlusion: A) Selective occlusion of segmental portal vein by a balloon introduced under ultrasonographic guidance. The arrows show (more ...) |
The majority of liver resections can be done with no clamping at all.30,31 In some patients total hepatic vascular isolation is needed. This isolation can be partial (meaning occlusion of the inflow and only one hepatic vein32,33) or total (meaning occlusion of the inflow). Outflow occlusion is obtained by occluding the vena cava above and below the hepatic veins34–37 (Figure 3). In cases of a small tumor adherent to a hepatic vein, isolation of the corresponding liver by clamping the inflow and the hepatic vein can facilitate and render surgery safe if the lesion is resected with the adherent vein. When a large tumor is found to have entered the vena cava, this technique enables bloodless resection of the involved vena cava and safe reconstruction of its continuity.38,39 Total vascular exclusion (TVE) of the liver can be applied safely for as long as 60 minutes. This can be extended to 8 hours by using hypothermia (Figure 4), as is done in liver transplantation. The liver resection can be done in situin vivo as first described by Fortner et al.,40–42ex situ in vivo as described by Hannoun et al.,42–45 or ex situ ex vivo as described by Pichlmayr et al.46 (Figure 5, next page). This innovative approach to liver resection has a high rate of complication and even mortality. 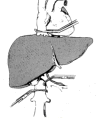 | Figure 3 Total vascular exclusion of the liver by clamping the infrahepatic and suprahepatic inferior vena cava and the hepatoduodenal ligament. |
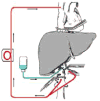 | Figure 4Total vascular exclusion of the liver with hypothermia as described by Fortner et al. 40 The liver is excluded (as in Figure 3). Veno-venous bypass of the liver is performed (red lines), and hypothermic solution is infused into the portal vein (in blue). (more ...) |
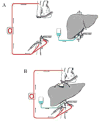 | Figure 5Total vascular exclusion for complex liver resections. A) The ex-situex-vivo technique described by Pichlmayr et al. 46 B) The ex-situin-vivo technique described by Hannoun et al. 43 |
Another concept in liver surgery that guides surgeons toward safe surgery is that “the liver is parenchyma tissue with blood vessels inside”. The rationale behind this concept is to dissect this parenchyma tissue while ensuring hemostasis. The concept was popularized following the development of new devices that enabled such dissection, e.g. the harmonic scalpel,47–49 LigaSure,50–52 tissue link,53 radiofrequency,54,55 and the “Habib sealer”.56,57 These devices allow the parenchyma to be cut without having to clamp the pedicle. Nevertheless, the clamp crushing technique is still widely used.58–61
The main indication for liver resection today is liver metastasis resulting from advanced cancers of the colon and rectum. Apart from liver surgery for trauma or hepatocellular carcinoma in cirrhotic patients where the mortality is high,62 the overall operative mortality in liver resections is between 0% and 2%.63,64 Trained liver surgery teams can achieve less than 1% mortality. This is a great advance in comparison to the mortality in liver surgery in early reports, which reached a mortality rate as high as 20%.65 |
1. Langenbuch C. Ein Fall von Resecktion eines linksseitigen Schnurlappens der Leber. Berl Klin Wochenschr. 1888;25:37. German. 4. Lortat-Jacob JL, Robert HG. Well defined technic for right hepatectomy. Presse Med. 1952;60:549–51. French. 5. Couinaud C. Lobes et segments hépatiques. Presse Med. 1954;62:709. French. 6. Couinaud, C. Le foie. Paris: Masson; 1957. French. 7. Stellamor K. [About the advantages of the sonographic diagnostic in the field of the radiologist (author’s transl)]. Rontgenblatter. 1976;29:544–52. German. 8. Gosink BB. Evaluation of hepatic neoplasms. AJR AM J Roentgenol. 1980;134:621. 9. Bismuth H. Surgical anatomy and anatomical surgery of the liver. World J Surg. 1982;6:3–9. doi:10.1007/BF01656368.10. Makuuchi M, Hasegawa H, Yamazaki S. Intraoperative ultrasonic examination for hepatectomy. Ultrasound Med Biol. 1983;(Suppl 2):493–7. 11. Makuuchi M, Hasegawa H, Yamazaki S. Development on segmentectomy and subsegmentectomy of the liver due to introduction of ultrasonography. Nippon Geka Gakkai Zasshi. 1983;84:913–7. Japanese. 12. Bismuth H, Castaing D. Peroperative echography in hepatobiliary surgery. Ann Gastroenterol Hepatol (Paris). 1984;20:221–3. French. 13. Bismuth H, Castaing D, Kunstlinger F. Peroperative echography in hepatobiliary surgery. Presse Med. 1984;13:1819–22. French. 14. Bismuth H, Houssin D, Castaing D. Major and minor segmentectomies “réglées” in liver surgery. World J Surg. 1982;6:10–24. doi:10.1007/BF01656369.15. Neuhaus W. [Intraoperative manometry and roentgenography of biliary system and its significance for surgery of the bile ducts]. Langenbecks Arch Klin Chir Ver Dtsch Z Chir. 1953;275:395–403. doi:10.1007/BF01438628.16. Lin TY, Tsu K, Mien C, Chen C. Study on lobectomy of the liver. J Formosa Med Assoc. 1958;57:742–9. 19. Pamecha V, Gurusamy KS, Sharma D, Davidson BR. Techniques for liver parenchymal transection: a meta-analysis of randomized controlled trials. HPB (Oxford). 2009;11:275–81. 20. Rau HG, Wichmann MW, Schinkel S, et al. [Surgical techniques in hepatic resections: Ultrasonic aspirator versus Jet-Cutter. A prospective randomized clinical trial]. Zentralbl Chir. 2001;126:586–90. doi:10.1055/s-2001-16573. German. 22. Man K, Fan ST, Ng IO, Lo CM, Liu CL, Wong J. Prospective evaluation of Pringle maneuver in hepatectomy for liver tumors by a randomized study. Ann Surg. 1997;226:704–13. doi:10.1097/00000658-199712000-00007.23. Man K, Fan ST, Ng IO, et al. Tolerance of the liver to intermittent pringle maneuver in hepatectomy for liver tumors. Arch Surg. 1999;134:533–9. doi:10.1001/archsurg.134.5.533.24. Lazorthes F, Chiotasso P, Chevreau P, Materre JP, Roques J. Hepatectomy with initial suprahilar control of intrahepatic portal pedicles. Surgery. 1993;113:103–8. 25. Launois B, Jamieson GG. The posterior intrahepatic approach for hepatectomy or removal of segments of the liver. Surg Gynecol Obstet. 1992;174:155–8. 26. Figueras J, Lopez-Ben S, Llado L, et al. Hilar dissection versus the “glissonean” approach and stapling of the pedicle for major hepatectomies: a prospective, randomized trial. Ann Surg. 2003;238:111–9. doi:10.1097/000006582003070 00 00015.27. Shimamura Y, Gunven P, Takenaka Y, et al. Selective portal branch occlusion by balloon catheter during liver resection. Surgery. 1986;100:938–41. 28. Castaing D, Garden OJ, Bismuth H. Segmental liver resection using ultrasound-guided selective portal venous occlusion. Ann Surg. 1989;210:20–3. doi:10.1097/00000658-198907000-00003.29. Bismuth H, Houssin D, Castaing D. Major and minor segmentectomies “reglees” in liver surgery. World J Surg. 1982;6:10–24. doi:10.1007/BF01656369.30. Capussotti L, Muratore A, Ferroro A, Massucco P, Ribero D, Polastri R. Randomized clinical trial of liver resection with and without hepatic pedicle clamping. Br J Surg. 2006;93:685–9. doi:10.1002/bjs.5301.31. Nuzzo G, Giuliante F, Giovannini I, Vellone M, De Cosmo G, Capelli G. Liver resections with or without pedicle clamping. Am J Surg. 2001;181:238–46. doi:10.1016/S0002-9610(01)00555-4.32. Smyrniotis VE, Kostopanagiotou GG, Contis JC, et al. Selective hepatic vascular exclusion versus Pringle maneuver in major liver resections: prospective study. World J Surg. 2003;27:765–9. doi:10.1007/s00268-003-6978-8.33. Cherqui D, Malassagne B, Colau PI, Brunetti F, Rotman N, Fagniez PL. Hepatic vascular exclusion with preservation of the caval flow for liver resections. Ann Surg. 1999;230:24–30. doi:10.1097/00000658-199907000-00004.34. Huguet C, Gallot D, Offenstadt G, Coloigner M. [Total vascular exclusion of the liver in extensive hepatic exeresis. Value and limits]. Nouv Presse Med. 1976;5:1189–92. French. 36. Offenstadt G, Huguet C, Gallot D, Bloch P. Hemodynamic monitoring during complete vascular exclusion for extensive hepatectomy. Surg Gynecol Obstet. 1978;146:709–13. 37. Huguet C, Nordlinger B, Galopin JJ, Bloch P, Gallot D. Normothermic hepatic vascular exclusion for extensive hepatectomy. Surg Gynecol Obstet. 1978;147:689–93. 38. Maeba T, Okano K, Mori S, et al. Retrohepatic vena cava replacement of hepatic malignancies without using total hepatic vascular exclusion or extracorporeal bypass. Hepatogastroenterology. 2001;48:1455–60. 39. Ohwada S, Kawashima Y, Ogawa T, et al. Extended hepatectomy with ePTFE graft vena caval replacement and hepatic vein reconstruction: a case report. Hepatogastroenterology. 1999;46:1151–5. 41. Azoulay D, Eshkenazy R, Andreani P, et al. In situ hypothermic perfusion of the liver versus standard total vascular exclusion for complex liver resection. Ann Surg. 2005;241:277–85. doi:10.1097/01.sla.0000152017.62778.2f.42. Delrivière L, Hannoun L. In situ and ex situ procedures for complex major liver resections requiring prolonged hepatic vascular exclusion in normal and diseased livers. J Am Coll Surg. 1995;181:272–6. 43. Hannoun L, Balladur P, Delva E, et al. “Ex situ-in vivo” surgery of the liver: a new technique in liver surgery. Principles and preliminary results. Gastroenterol Clin Biol. 1991;15:758–61. French. 44. Hannoun L, Borie D, Balladur P, et al. [Ex situ-in vivo hepatic resection. Technique and initial results]. Chirurgie. 1992;118:292–6. discussion 296–7. French. 46. Pichlmayr R, Grosse H, Hauss J, et al. Technique and preliminary results of extracorporeal liver surgery (bench procedure) and of surgery of the in situ perfused liver. Br J Surg. 1990;77:21–6. doi:10.1002/bjs.1800770107.47. Gertsch P, Pelloni A, Guerra A, Krpo A. Initial experience with the harmonic scalpel in liver surgery. Hepatogastroenterology. 2000;47:763–6. 48. Schmidbauer S, Hallfeldt KK, Sitzmann G, Kantelhardt T, Trupka A. Experience with ultrasound scissors and blades (UltraCision) in open and laparoscopic liver resection. Ann Surg. 2002;235:27–30. doi:10.1097/00000658-200201000-00004.49. Aldrighetti L, Pulitanò C, Arru M, Catena M, Finazzi R, Ferla G. “Technological” approach versus clamp crushing technique for hepatic parenchymal transection: a comparative study. J Gastrointest Surg. 2006;10:974–9. doi:10.1016/j.gassur.2006.02.002.50. Saiura A, Yamamoto J, Koga R, Seki M, Yamaguchi T. Liver transection using the LigaSure sealing system. HPB (Oxford). 2008;10:239–43. 51. Ikeda M, Hasegawa K, Sano K, et al. The vessel sealing system (LigaSure) in hepatic resection: a randomized controlled Trial. Ann Surg. 2009;250:199–203. doi:10.1097/SLA.0b013e3181a334f9.52. Nanashima A, Tobinaga S, Abo T, Nonaka T, Sawai T, Nagayasu T. Usefulness of the combination procedure of crash clamping and vessel sealing for hepatic resection. J Surg Oncol. 2010;102:179–83. doi:10.1002/jso.21575.53. Poon RT, Fan ST, Wong J. Liver resection using a saline-linked radiofrequency dissecting sealer for transection of the liver. J Am Coll Surg. 2005;200:308–13. doi:10.1016/j.jamcollsurg.2004.10.008.55. Poon RT, Ng KK, Lam CM, et al. Learning curve for radiofrequency ablation of liver tumors: prospective analysis of initial 100 patients in a tertiary institution. Ann Surg. 2004;239:441–9. doi:10.1097/01.sla.0000118565.21298.0a.56. Weber JC, Navarra G, Jiao LR, Nicholls JP, Jensen SL, Habib NA. New technique for liver resection using heat coagulative necrosis. Ann Surg. 2002;236:560–3. doi:10.1097/00000658200211000-00004.57. Ferko A, Lesko M, Subrt Z, et al. A modified radiofrequency-assisted approach to right hemihepatectomy. Eur J Surg Oncol. 2006;32:1209–11. doi:10.1016/j.ejso.2006.07.013.58. Jarnagin WR, Gonen M, Fong Y, et al. Improvement in perioperative outcome after hepatic resection: analysis of 1,803 consecutive cases over the past decade. Ann Surg. 2002;236:397–406. doi:10.1097/00000658-200210000-00001.59. Imamura H, Seyama Y, Kokudo N, et al. One thousand fifty-six hepatectomies without mortality in 8 years. Arch Surg. 2003;138:1198–206. doi:10.1001/archsurg.138.11.1198.60. Sun HC, Qin LX, Lu L, et al. Randomized clinical trial of the effects of abdominal drainage after elective hepatectomy using the crushing clamp method. Br J Surg. 2006;93:422–66. doi:10.1002/bjs.5260.61. Lesurtel M, Selzner M, Petrowsky H, McCormack L, Clavien PA. How should transection of the liver be performed? A prospective randomized study in 100 consecutive patients: comparing four different transection strategies. Ann Surg. 2005;242:814–22. doi:10.1097/01.sla.0000189121.35617.d7.63. Asiyanbola B, Chang D, Gleisner AL, et al. Operative mortality after hepatic resection: are literature-based rates broadly applicable? J Gastrointest Surg. 2008;12:842–51. doi:10.1007/s11605-008-0494-y.64. Dimick JB, Cowan JA Jr, Knol JA, Upchurch GR Jr. Hepatic resection in the United States, indications, outcomes, and hospital procedural volumes from a nationally representative database. Arch Surg. 2003;138:185–91. doi:10.1001/archsurg.138.2.185.65. Almersjö O, Bengmark S, Hafström L. Liver resection for cancer. Acta Chir Scand. 1976;142:139–44. 66. Houssin D, Franco D, Berthelot P, Bismuth H. Heterotopic liver transplantation in end-stage HBsAg-positive cirrhosis. Lancet. 1980;1:990–3. doi:10.1016/S0140-6736(80)91435-X.67. Bismuth H, Houssin D. Reduced-sized orthotopic liver graft in hepatic transplantation in children. Surgery. 1984;95:367–70. 68. Brölsch CE, Stevens LH, Whitington PF. The use of reduced-size liver transplants in children, including split livers and living related liver transplants. Eur J Pediatr Surg. 1991;1:166–71. doi:10.1055/s-2008-1042480.69. Azoulay S, Samuel D, Castaing D, et al. Domino liver transplants for metabolic disorders: experience with familial amyloidotic polyneuropathy. J Am Coll Surg. 1999;189:584–93. doi:10.1016/S1072-7515(99)00208-2.70. Figueras J, Parés D, Munar-Ques M, et al. Experience with domino or sequential liver transplantation in patients with familial amyloidotic polyneuropathy. Transplant Proc. 2002;34:307–8. doi:10.1016/S0041-1345(01)02776-2.71. Bismuth H, Samuel D, Castaing D, et al. Orthotopic liver transplantation in fulminant and subfulminant hepatitis. The Paul Brousse experience. Ann Surg. 1995;222:109–19. doi:10.1097/00000658-199508000-00002.72. Bismuth H, Morino M, Castaing D, et al. Emergency orthotopic liver transplantation in two patients using one donor liver. Br J Surg. 1989;76:722–4. doi:10.1002/bjs.1800760723.73. Azoulay D, Astarcioglu I, Bismuth H, et al. Split-liver transplantation. The Paul Brousse policy. Ann Surg. 1996;224:737–46. doi:10.1097/00000658-199612000-00009. discussion 746–8. 74. Bismuth H, Azoulay D, Samuel D, et al. Auxiliary partial orthotopic liver transplantation for fulminant hepatitis. The Paul Brousse experience. Ann Surg. 1996;224:712–24. doi:10.1097/00000658-199612000-00007. discussion 724–6. 75. Adam R, Avisar E, Ariche A, et al. Five-year survival following hepatic resection after neoadjuvant therapy for nonresectable colorectal. Ann Surg Oncol. 2001;8:347–53. doi:10.1007/s10434-001-0347-3.76. Bismuth E, Adam R, Lévi F, et al. Resection of nonresectable liver metastases from colorectal cancer after neoadjuvant chemotherapy. Ann Surg. 1996;224:509–20. doi:10.1097/00000658-199610000-00009.77. Adam R, Delvart V, Pascal G, et al. Rescue surgery for unresectable colorectal liver metastases downstaged by chemotherapy: a model to predict long term survival. Ann Surg. 2004;240:644–57. discussion 657–8. 78. Folprecht G, Gruenberger T, Bechstein WO, et al. Tumour response and secondary resectability of colorectal liver metastases following neoadjuvant chemotherapy with cetuximab: the CELIM randomised phase 2 trial. Lancet Oncol. 2010;11:38–47. doi:10.1016/S1470-2045(09)70330-4.79. Min BS, Kim NK, Ahn JB, et al. Cetuximab in combination with 5-fluorouracil, leucovorin and irinotecan as a neoadjuvant chemotherapy in patients with initially unresectable colorectal liver metastases. Onkologie. 2007;30:637–43. doi:10.1159/000109957.80. Pozzo C, Barone C, Kemeny NE. Advances in neoadjuvant therapy for colorectal cancer with liver metastases. Cancer Treat Rev. 2008;34:293–301. doi:10.1016/j.ctrv.2008.01.004.81. Kawasaki S, Makuuchi M, Kakazu T, et al. Resection for multiple metastatic liver tumors after portal embolization. Surgery. 1994;115:674–7. 82. Madoff D, Abdalla E, Vauthey J. Portal vein embolization in preparation for major hepatic resection: evolution of a new standard of care. J Vasc Interv Radiol. 2005;16:779–90. 83. Elias D, Santoro R, Ouellet J, Osmak L, de Baere T, Roche A. Simultaneous percutaneous right portal vein embolization and left liver tumor radiofrequency ablation prior to a major right hepatic resection for bilateral colorectal metastases. Hepatogastroenterology. 2004;51:1788–91. 85. Elias D, Ouellet J, De Baere T, Lasser P, Roche A. Preoperative selective portal vein embolization before hepatectomy for liver metastases: long-term results and impact on survival. Surgery. 2002;131:294–9. doi:10.1067/msy.2002.120234.86. Azoulay D, Castaing D, Smail A, et al. Resection of nonresectable liver metastases from colorectal cancer after percutaneous portal vein embolization. Ann Surg. 2000;231:480–6. doi:10.1097/00000658-200004000-00005.87. Shimada H, Tanaka K, Masui H, et al. Results of surgical treatment for multiple (>or=5 nodules) bi-lobar hepatic metastases from colorectal cancer. Langenbecks Arch Surg. 2004;389:114–21. doi:10.1007/s00423-003-0447-6.88. Akasu T, Moriya Y, Takayama T. A pilot study of multimodality therapy for initially unresectable liver metastases from colorectal carcinoma: hepatic resection after hepatic arterial infusion chemotherapy and portal embolization. Jpn J Clin Oncol. 1997;27:331–5. doi:10.1093/jjco/27.5.331.89. Broering D, Hillert C, Krupski G, et al. Portal vein embolization vs. portal vein ligation for induction of hypertrophy of the future liver remnant. J Gastrointest Surg. 2002;6:905–13. doi:10.1016/S1091-255X(02)00122-1.90. Takayama T, Makuuchi M. Preoperative portal vein embolization: is it useful? J Hepatobiliary Pancreat Surg. 2004;11:17–20. doi:10.1007/s00534-002-0800-0.91. Abdalla EK, Barnett CC, Doherty D, Curley SA, Vauthey JN. Extended hepatectomy in patients with hepatobiliary malignancies with and without preoperative portal vein embolization. Arch Surg. 2002;137:675–80. doi:10.1001/archsurg.137.6675.92. Wakabayashi H, Ishimura K, Okano K, et al. Application of preoperative portal vein embolization before major hepatic resection in patients with normal or abnormal parenchyma. Surgery. 2002;131:26–33. doi:10.1067/msy.2002.118259.93. Adam R, Lucidi V, Bismuth H. Hepatic colorectal metastases: methods of improving resectability. Surg Clin North Am. 2004;84:659–71. doi:10.1016/j.suc.2003.12.005.94. Goere D, Farges O, Leporrier J, Sauvanet A, Vilgrain V, Belghiti J. Chemotherapy does not impair hypertrophy of the left liver after right portal vein obstruction. J Gastrointest Surg. 2006;10:365–70. doi:10.1016/j.gassur.2005.09.001.95. Beal I, Anthony S, Papadopoulou A, et al. Portal vein embolization prior to hepatic resection for colorectal liver metastases and the effects of periprocedure chemotherapy. Br J Radiol. 2006;79:473–8. doi:10.1259/bjr/29855825.96. Jaeck D, Bachellier P, Nakano H, et al. One or two-stage hepatectomy combined with portal vein embolization for initially nonresectable colorectal liver metastases. Am J Surg. 2003;185:221–9. doi:10.1016/S0002-9610(02)01373-9.97. Adam R, Laurent A, Azoulay D, Castaing D, Bismuth H. Two stage hepatectomy: a planned strategy to treat unresectable liver tumors. Ann Surg. 2000;232:777–85. doi:10.1097/00000658-2000 12000-00006.98. Jaeck D, Oussoultzoglou E, Rosso E, Greget M, Weber JC, Bachellier P. A two-stage hepatectomy procedure combined with portal vein embolization to achieve curative resection for initially unresectable multiple and bilobar colorectal liver metastases. Ann Surg. 2004;240:1037–51. doi:10.1097/01.sla.0000145965.86383.89.99. Nguyen KT, Laurent A, Dagher I, et al. Minimally invasive liver resection for metastatic colorectal cancer: a multi-institutional, international report of safety, feasibility, and early outcomes. Ann Surg. 2009;250:842–8. doi:10.1097/SLA.0b013e3181bc789c.103. Vibert E, Perniceni T, Levard H, Denet C, Shahri NK, Gayet B. Laparoscopic liver resection. Br J Surg. 2006;93:67–72. doi:10.1002/bjs.5150.105. Gumbs AA, Gayet B, Gagner M. Laparoscopic liver resection: when to use the laparoscopic stapler device. HPB (Oxford). 2008;10:296–303. 106. Bryant R, Laurent A, Tayar C, Cherqui D. Laparoscopic liver resection-understanding its role in current practice: the Henri Mondor Hospital experience. Ann Surg. 2009;250:103–11. doi:10.1097/SLA.0b013e3181ad6660.107. Soubrane O, Cherqui D, Scatton O, et al. Laparoscopic left lateral sectionectomy in living donors: safety and reproducibility of the technique in a single center. Ann Surg. 2006;244:815–20. doi:10.1097/01.sla.0000218059.31231.b6. |
