In mainland China, CT was often a first-line investigation for COVID-19, and some hospitals dedicated specific CT scanners for examining suspected COVID-19 patients only.7 This strategy may be logical for an endemic population in which the main load of hospitalized patients is suspected or diagnosed with COVID-19; however, in a cosmopolitan region, it poses great logistic difficulty on the radiology department and presents a potential hazard of cross-infecting personnel as well as other patients. For these reasons, the American College of Radiology suggested using portable CXR as the first imaging tool,8 and both Italian and British hospitals shifted to that direction.7,9 Israel’s National Center for Information and Knowledge in the Battle Against the Coronavirus has not recommended the use of imaging, including CT, for diagnosing COVID-19, but rather has relied on laboratory diagnoses,10 with imaging reserved for monitoring of the disease in confirmed patients.
Chest X-ray
Chest X-rays are an effective bedside modality through the use of portable devices. The classic CXR findings in COVID-19 are areas with increased hazy opacities and/or bilateral consolidations, usually with distribution in the lower lobes.
10–12 Pleural effusion is uncommon (
Figure 1). The diagnostic sensitivity of CXR in the early stage of the disease is about 69%,
7 although the specificity is unknown.
 | Figure 1 Chest X-ray (CXR) of a Confirmed COVID-19 Patient in the Sitting Position |
Computed Tomography
The CT findings in COVID-19 are well documented. Ground-glass opacification (GGO; defined as hazy increased lung attenuation with preservation of bronchial and vascular margins) and consolidative pulmonary opacities (defined as opacification with obscuration of margins of vessels and airway walls), often with a bilateral and peripheral lung distribution, are the hallmarks of COVID-19 patients’ chest CT images (
Figure 2).
13,14 | Figure 2 Chest Computed Tomography (CT) of a Confirmed COVID-19 Patient |
Bernheim et al. correlated the CT findings of 94 confirmed COVID-19 patients (based on positive RT-PCR) with the infection duration (early phase: days 0–2; intermediate phase at days 2–5; and late phase: days 6–12). During the first two days of the disease, 56% of patients had a normal CT as compared to 4% in the late phase. Early findings showed a bilateral distribution of small GGOs, mainly peripherally located. Over time, opacifications and consolidations increased, additional patterns such as crazy paving, a reversed halo sign, and linear opacities were noted (Figure 3), and the peripheral predilection and multilobular distribution became more apparent.14 Jin et al. recognized five temporal stages classified as ultra-early, early, rapid progression, consolidation, and dissipation stages. In the dissipation stage, lesions may be reduced in number and extension, with small ill-defined interlobular septal thickening remaining.15 These findings were in concordance with other publications, summarized in a review by Lomoro et al.12
 | Figure 3 Chest CT Showing the Temporal Stages of COVID-19 Infection (Findings Indicated by Red Frame) |
The role of CT for COVID-19 diagnosis merits exploration. In endemic areas in China, due to the shortcomings of RT-PCR, CT was used for early disease detection.7 Ai et al. showed that CT has greater sensitivity than RT-PCR for COVID-19 diagnosis in a highly endemic area and high pre-test probability for disease.16 Bernheim, on the other hand, showed that during the first few days of infection more than half of the patients had a normal CT, and concluded that CT had limited sensitivity and a negative predictive value early after symptom onset, and that it was therefore not a reliable standalone tool for ruling out COVID-19 infection.14 Lomoro et al. showed normal CT studies for almost 5% of confirmed COVID-19 patients.12
Another important question regarding CT is whether or not it can differentiate between COVID-19 and other viral pneumonias. The CT findings in different virus families have been well documented, but an overlap of imaging findings and atypical presentation might complicate their interpretation.17 In addition, other processes such as influenza pneumonia and organizing pneumonia may manifest as bilateral peripheral GGOs.18 Bai et al. retrospectively compared the CT findings in COVID-19 to other viral pneumonias. They found a tendency toward peripheral rather than central pathology, mainly GGOs, fine reticular opacities, vascular thickening, with a lower frequency of pleural effusion and lymphadenopathy in COVID-19 as compared to other viral pneumonias. Radiologists using CT to differentiate between COVID-19 and non-COVID-19 pneumonia did so with moderate sensitivity (80.4%) and high specificity (96.6%), thus CT appears to be better suited to ruling out COVID-19.19
Ultrasound
Lung and chest US are already being used by intensive care physicians as a bedside tool for evaluating a variety of pathologies including pneumothorax, pleural effusion, acute dyspnea, pulmonary edema, pulmonary embolism, pneumonia, interstitial processes, and for patients on mechanical ventilatory support.
20 In the early days of the COVID-19 outbreak, clinicians were originally the ones who performed lung US on infected patients and as a result mastered the technology; they subsequently published their findings.
21,22
Any recent-generation US device can be used for lung exams: premium devices, small portable ones, and even wireless US devices are all adequate for lung and thorax imaging. Different probes are used, mainly curvilinear probes with an abdominal preset for deeper and larger lung field coverage, and linear probes for better visualization of the specific superficial pathology. Phased array probes designed for cardiac imaging may also be used.20 The operator should be well trained and familiar with both the machine and the technique.23 The operator’s goal is to cover as much as possible of the lungs in a systematic, quick, and efficient manner. For reproducibility and uniformity of reporting, the lungs are scanned at fixed points. Most of the COVID-19 publications relating to lung US used a focused 12-area approach. The thorax was scanned bilaterally (Right 1–6 and Left 1–6) at the bedside: anterior superior and inferior, lateral superior and inferior, posterior superior and inferior.21–26
Any lung pathology that extends to the pleural surface may be visualized with US. However, US cannot detect lesions deep within the lung, since the aerated lung blocks its transmission.
Normal chest sonography will show the detailed chest wall anatomy including the skin, subcutaneous tissue, muscular layer, and ribs. The pleura slides during respiration and is seen as a sharp continuous white line, posterior and adjacent to the chest wall. In addition to blocking US transmission, the aerated lung is hypoechoeic and homogenic. A reverberation artifact, generated due to the interphase between the superficial layers and the air in the lungs, creates what are known as white A-lines. These A-lines appear in constant spacing, parallel to the pleural line and to each other, and fade gradually with depth (Figure 4).
In all lung pathologies, the A-line artifacts are lost and a different artifact appears, as the in B-lines, which are generated when the interstitium or small number of subpleural alveoli fill with fluid. The interphase between the aerated areas and the fluid-filled areas create the B-line artifact. If present, these longitudinal B-lines begin at the pleural line and run without fading to the deeper field. Their sharpness, number, distribution, and location differ between lung pathologies and constitute clues to diagnosis. Regardless of the specific pathology, the decay of B-lines and the reappearance of A-lines are considered to be reliable signs of lung recovery.24
Small subpleural consolidations are seen in US as hypoechoic areas with or without air bronchogram and local loss of the pleural line. A proper segmental to lobar consolidation with complete loss of the aerated lung parenchyma is visualized as a solid tissue, with a strong resemblance to the liver parenchyma, known as hepatization of the lung. A dynamic air bronchogram is seen within the consolidated lung.
Only a few publications, mainly case reports or small series, have described the sonographic findings in COVID-19. Classical findings include an irregular pleural line with small subpleural consolidations, disappearance of the normal A-lines, and the appearance of multiple B-lines. The B-lines have areas that are fused (waterfall sign) or diffused (white lung sign). The B-lines noted in COVID-19 are more likely to be fused, fixed, and with blurred edges than those appearing with cardiogenic pulmonary edema. Also noted are small subpleural multifocal, non-translobar, and translobar consolidations in a variety of patterns, with occasional mobile air bronchograms. Pleural effusion in COVID-19 is uncommon, and the lesions are mostly located in the posterior fields of both lungs. Spared areas are present bilaterally, mixed with pathological areas (Figure 5). With COVID-19 recovery, the A-lines reappear, replacing the B-lines.22
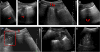 | Figure 5 Ultrasound of a COVID-19 Lung Injury |
The use of lung US to evaluate several respiratory conditions has been widely documented.20 Lung US provides results similar to chest CT and superior to standard chest radiography for evaluation of pneumonia and/or adult respiratory distress syndrome (ARDS).20 Several studies have noted a good correlation between lung US and CT for COVID-19.12,21,22,25,26 In addition, Yang et al. showed an improved sensitivity of lung US compared to CT in 29 patients with COVID-19 pneumonia.26
Another advantage of lung US is the ability of clinicians to perform this procedure at the bedside. In the presence of a highly contagious pathogen such as COVID-19, the use of a single dedicated bedside US device reduces the chance of cross-infection. Since no ionizing radiation is involved, the study may be repeated as clinically needed. The 12-area method allows for precise reporting and proper follow-up. Sterilization of portable US devices, especially the smaller or wireless machines, is considerably easier and faster as compared to a CT machine. Finally, portable US machines are relatively inexpensive and can be purchased for any hospital department as well as any community clinic responsible for treating COVID-19 patients outside of the hospital setting.
